

Sadiel Triana
Question 83: Wet flue gas scrubber NOx removal technology often results in excessive nitrates in the purge water. What are refiners doing to limit nitrates going to their wastewater treatment plant?
GIM (Technip Stone & Webster)
Nitrates and nitrites are inevitable byproducts from the removal of NOx from FCC flue gas. Obviously, one way to lower this nitrate concentration going into the purge water is to reduce the NOx in the flue gas, which we discussed a couple of questions ago. So, assuming that we have done everything possible to reduce the NOx in your flue gas, we will then need to look at 1) the impact of nitrates in the wastewater treatment system and 2) some of the solutions available to address the increase of nitrate in the purge stream to the wastewater treatment plant. After I go through this formal response, I think it will be helpful to open up the floor for feedback of any experiences you all may have. It will be a great way to address this particular question in more detail.
The incremental introduction of nitrates can bring imbalance to the existing wastewater treatment plant, which has its own set of capacity limitations, peaks, and valleys. There are several options and practices for pre-treating this purge stream ahead of the wastewater treatment plant, which becomes very important; because in some areas of the country, more stringent NOx regulations are in effect. Additional purge water will increase effluent sodium concentration. Also, the increase in nitrate concentration will increase the likelihood of higher nitrification in the secondary clarifier, which may result in exceeding the chemical oxygen demand (COD) permit limits.

One refiner has addressed the increase in nitrate by modifying its existing aeration basin in the wastewater treatment plant. An anoxic (not anabolic) treatment system was used to metabolize the NaNO3 (sodium nitrate) by utilizing oxygen from the NaNO3 itself; hence, termed anoxic as opposed to aerobic or anaerobic.
An anoxic tank was used ahead of the refinery wastewater treatment plant to initially treat the wet gas scrubber purge stream. The sizing and treatment of the anoxic tank is heavily dependent on the temperature of the purge water, as well as the size of the existing wastewater treatment plant and its permit limits. In this specific example, a 75% removal of the nitrate in the purge stream was achieved.
There are a couple of other smaller-scale industrial application options. Ion exchange is one of the technologies used to remove nitrates. Nitrate ions are exchanged with other ions, like chlorides, through a resin media. The benefit of this is that it is fairly independent of the concentration and temperature of the purge water, unlike the previous example I just mentioned. Simple distillation processes can also be incorporated ahead of the wastewater treatment plant to concentrate the nitrates and other minerals and send the treated water downstream.

ROBERTSON (AFPM)
That is the response for this question. Are there any other comments or questions from the audience? This question was a little exotic in its nature, so we are hoping that some other operators or people will share their experiences.
MICHAEL WARDINSKY (Phillips 66)
I am curious to know if this issue of having to revamp your wastewater plant has shifted anyone’s flue gas treatment options back to an SCR (selective catalytic reduction) or SNCR (selective non-catalytic reduction) versus a technology like LoTOx™ where you take the NOx, convert it into nitrate, and then put it in the wastewater. Did that change anyone’s approach to removing NOx from the flue gas?
BULL (Valero Energy Corporation)
We have various LoTOx™ applications, which were just taken into account during the design phase. We did not have to add equipment after startup. So as far as the capital cost for the full project, it did not have a significant impact.
STEVE GIM (Technip Stone & Webster)
Source of Nitrate: Nitrates and nitrites are inevitable byproducts from the removal of NOx from the FCC flue gas. One obvious way to reduce the number of nitrates is to reduce the source of NOx. This subject has been extensively discussed in many of the previous FCC panels. This question was not chosen from the original list. Some of the main parameters are combustion kinetics in the regenerator (excess oxygen level, temperatures, promoters, and CO level), FCC hardware (regenerator designs, air/catalyst distribution, and stripper efficiency), and, to some degree, the nitrogen content of the fresh feed to FCCs. We will cover the nitrates produced from the wet gas scrubber, assuming you have done everything you can to control the source of NOx. I appreciate our colleagues from Belco for their contribution of additional input addressing some of the salient issues surrounding the treatment of the nitrates.
Impact of Increase in Nitrate on Wastewater Treatment Plant: Incremental introduction of nitrates can bring imbalance to the existing wastewater treatment plant with its own set of capacity limitations and peaks and valleys. Technology options and practices for pre-treatment or rerouting of the purge water ahead of the wastewater treatment plant becomes important, especially in regions where more stringent NOx regulations are prevalent. Additional purge water will increase effluent sodium concentration. The increase in the nitrate concentration can also increase the likelihood of higher nitrification in the secondary clarifier, which may result in exceeding chemical oxygen demand (COD) permit limits.
Example of Nitrate Reduction Ahead of Wastewater Treatment Plant: One refiner addressed the increase in the nitrate by modifying his existing aeration basin in the water treatment plant to eliminate the problem. An anoxic (not anabolic) treatment system can be used to metabolize the NaNO3s by utilizing the oxygen from the NaNO3 itself; hence, the term anoxic (utilization of the inherent stream chemistry absent of an outside oxygen source), as opposed to aerobic or anaerobic.
An anoxic tank was used ahead of the refinery wastewater treatment plant to initially treat the wet gas scrubber purge stream. Anoxic tank sizing/treatment is very dependent on temperature. Every application would be different due to location and, even more importantly, the specific refineries wastewater treatment plant sizing, capabilities, and discharge permit limits. In this specific example, near 75% removal of nitrate was achieved.
In smaller scales, a couple of other options are available in other industrial application of nitrate removal. Ion exchange is one of the frequently used technologies for removal of nitrates. Nitrate ions are exchanged with other ions, like chlorides, through resin media. It is fairly independent of nitrate concentration and temperature of the purge water. A distillation process can also be incorporated to concentrate nitrates and other mineral contaminants, while sending the most the treated water downstream.
TINA SYVERT (Nalco Champion Energy Services)
Removal of nitrogen in refinery effluent water relies on biological processes. Nitrates can be handled by modifying a secondary treatment system with an anoxic reactor upstream of an aerobic reactor. The activated sludge-containing nitrates formed in the aerobic reactor as a result of the nitrification process (ammonia to nitrate) are returned to the anoxic zone where they are converted to nitrogen gas.
Question 84: What are the commercial experiences with low rare earth and zero rare earth SOx reduction additives? What are the incentives to return to traditional products now that rare earth prices have returned to historical levels?
KOEBEL (Grace Catalysts Technologies)
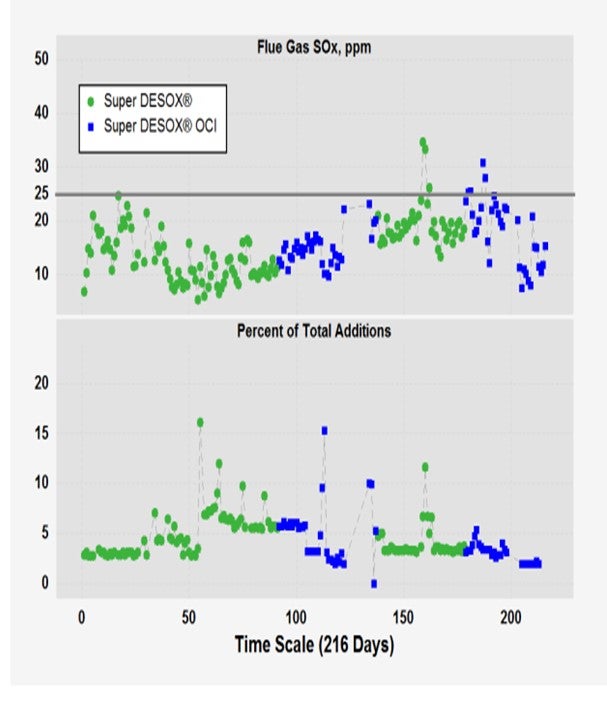
During the run-up in the cost of rare earth in 2011, Grace embarked on an expedited and extensive R&D (research and development) program to develop future generations of our Super DeSOx® additive to cut the rare earth content and try to minimize the financial impact that the ever-increasing cost of rare earth had on our customers’ cost of SOx compliance. The data I am showing on the slide is for our first-generation low-rare earth SOx additive called Super DeSOx®-OCI or optimized cerium input. This data is representative of a single trial, but I think it is reflective of our broader experience that the Super DeSOx®-OCI, with its lower cerium input, had an equivalent performance to that of traditional Super DeSOx®. The information presented here is the trial data on a pick-up factor basis of pounds of SOx removed from the flue gas divided by the total pounds of SOx additive used. You can see that the two additives are essentially equivalent.

The next slide shows the broader, expanded data from that six-month trial. You can see that the refinery switched back and forth between the two additives once or twice. On the percentage of daily addition basis, the SOx additive used was essentially at cost. The refiner was able to readily maintain its 25 ppm SOx emissions limit. So, we have had good experience with Super DeSOx®-OCI, our first-generation additive, and it has gained wide market acceptance.

As rare earth costs continued to escalate, we developed a second-generation low-rare earth SOx additive called Super DeSOx®-MCD or maximum cerium dispersion. Once we got the rare earth content down that low, there was a performance deficit for that additive. Generally, you had to add about 20% more additive. When rare earth was extremely expensive on a cost-per-day basis, adding a little more additive was well worth the cost. But as rare earth prices have now returned to historical levels, Super DeSOx®-OCI, our first-generation low-rare earth SOx additive, is our lowest cost-compliance additive.
The question was: What are the incentives for returning to your traditional Super DeSOx™ additives now that rare earth prices are low? Generally speaking, even though the performance is about the same, our Super DeSOx-OCI™ is a moderately lower-cost additive; so, we do not see a lot of incentive to return to the original Super DeSOx™ additive, except in very rare cases.
MARTIN EVANS (Johnson Matthey INTERCAT, Inc.)
When we introduced Super Soxgetter-II™ with about 8.0% rare earth, we were a little unsure what to expect. However, we found that as we gradually replaced all of the Super Soxgetter™ being used out there, overall the performance actually picked up in full-burn units by about 15%. That makes sense when you think about it. Consider full-burn units in which SOx additive efficiency is generally limited by the amount of magnesium that you can get into the units to pick up the SO2. When you take out the cerium, you end up replacing it with magnesium. Overall, the efficiency of Super Soxgetter-II™ actually ended up about 15% higher because of that slight increase in the magnesium content. It was not an effect we expected, but we were very pleased to see it. We do now also have Super Soxgetter-III™ out there, which has taken the cerium content down to 4%, and the performance remains about the same As Jeff mentioned, since the rare earth prices have declined right now, I think the incentive to go that low really is not there anymore. The manufacturing cost on that product is actually slightly higher; so today, it is not really an economic option for most people. But should rare earth prices increase again, we do have that product ready to go.
KEN BRUNO (Albemarle Corporation)
We continue to find that our zero rare earth products are highly effective in SOx reduction. In fact, our latest and newest innovation – SOxMaster-2™ – has been proven successful in several commercial trials. Again, please refer to the Answer Book for addition details.
JEFF KOEBEL (Grace Catalysts Technologies)
During the early phase of the run up in rare earth prices in 2011, Grace started an accelerated R&D effort to develop a new SuperDESOX® product with a lower rare earth content to minimize the financial impact on our customers. The first of the newly developed products was SuperDESOX®-OCI, or optimized cerium input. The effectiveness of these new additives can be measured by the SOx pickup factor, or PUF. This is defined as the pounds of SOx removed from the flue gas divided by the pounds of SOx additive used. In multiple commercial trials, SuperDESOX®-OCI proved to have equivalent PUFs to traditional SuperDESOX®. The total pounds of additive required to maintain a constant SOx emissions target is thus the same for both additives. Figure 1 illustrates in a commercial example where Super DESOX®-OCI has an equivalent pickup factor as does traditional Super DESOX®.

The refinery also maintained target SOx flue gas emission below the 25 ppm level (Figure 2). With its lower rare earth input, SuperDESOX®-OCI was clearly the lower cost option. This product gained wide market acceptance quickly.

As the cost of rare earth continued to climb, Grace developed SuperDESOX® MCD, or maximum cerium dispersion. This product further reduced the rare earth content of the additive to below the level of even SuperDESOX®-OCI. With rare earth inputs lowered so dramatically, there is a performance effect on the additive’s PUF; and generally speaking, it is necessary to add approximately 20% more SuperDESOX® MCD than SuperDESOX®-OCI or traditional SuperDESOX®. At the peak of the run up in rare earth prices, the required additional additive use was more than offset by the cost savings on a per pound basis, making SuperDESOX® MCD the lowest cost compliance option at the time.
Since that time, rare earth prices have significantly moderated and returned to mostly historical levels. However, Grace’s customers have generally not returned to using traditional SuperDESOX® simply because SuperDESOX®-OCI is a lower-cost product with equivalent performance. At this time, there is limited incentive to return to the use of typical first-generation “super” SOx additives.
RYAN NICKELL (Albemarle Corporation)
Albemarle continues to find that our zero rare earth products are highly effective for SOx reduction. Our newest innovation, SOxMASTER-2™, has proven effective in two commercial trials. In the first trial, this product effectively replaced an additive blend with cerium. In the second trial, a cerium-based additive was fully replaced at constant addition rate. SOxMASTER-2™ and the incumbent both showed a PUF of about 32 kg. SOx emission levels stayed constant. In addition, zero rare earth products are always the optimal choice in partial-burn applications.
With rare earth prices at historical levels, most refiners with full-burn units will still opt for cerium-based additives due to experience, availability, or unique requirements. However, in situations where refiners require high performance and are not able to use products containing cerium or rare earths, SOxMASTER-2™ is offered as a proven product.
Question 85: What are the advantages and disadvantages of catalyst mesoporosity [100 to 600 ǻngström (Å) diameter pores] in VGO (vacuum gas oil) operations?
KOEBEL (Grace Catalysts Technologies)
When talking about catalyst mesoporosity, we are referring to the matrix contribution, especially the 100 to 600 ǻngström pores found in the catalyst matrix activity. Catalyst matrix activity is really important, particularly in the Type I cracking depicted here, in the mix zone of the riser where the mix zone can be on the order of 1100°F. Even in VGO types of operations, you can have a significant portion of the total feed that may not vaporize at those conditions. Having effective diffusion is important for pre-cracking these types of feeds so that the zeolite can efficiently crack it further later on up the riser. Certainly, even in VGO operations, we think that avoiding diffusion limitations in the mix zone can be tremendously important.

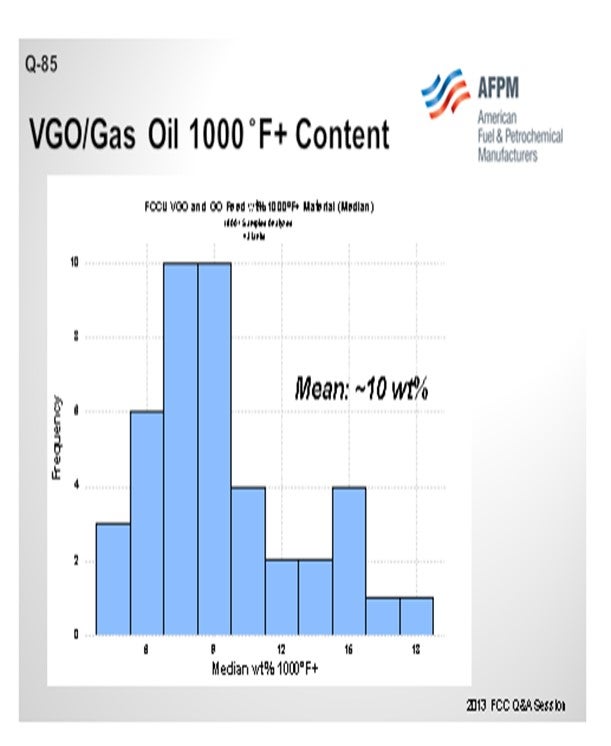
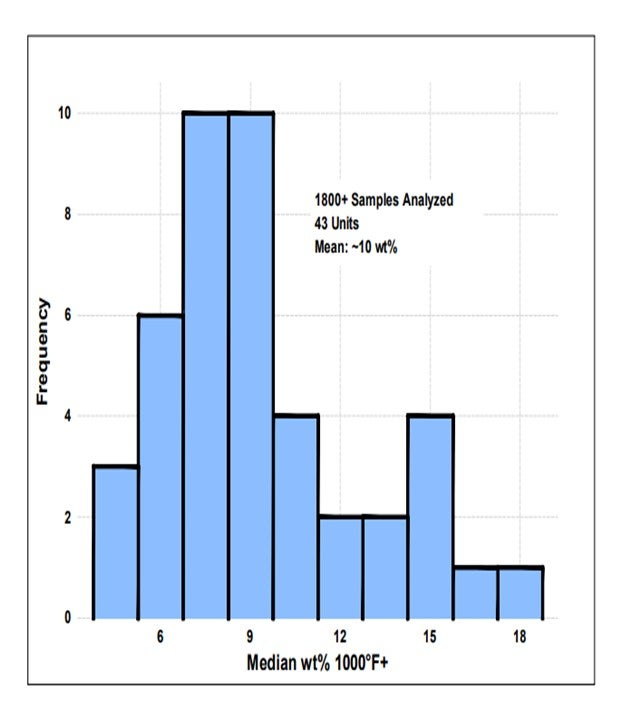
I queried the Grace database on FCC feed. Even in gas oil and VGO operations, the average was that about 10% of the feed boiled over at 1000°F; so clearly this is not just a resid phenomenon. This is a factor in FCC operation that Grace considers to be very important in even the VGO operations.
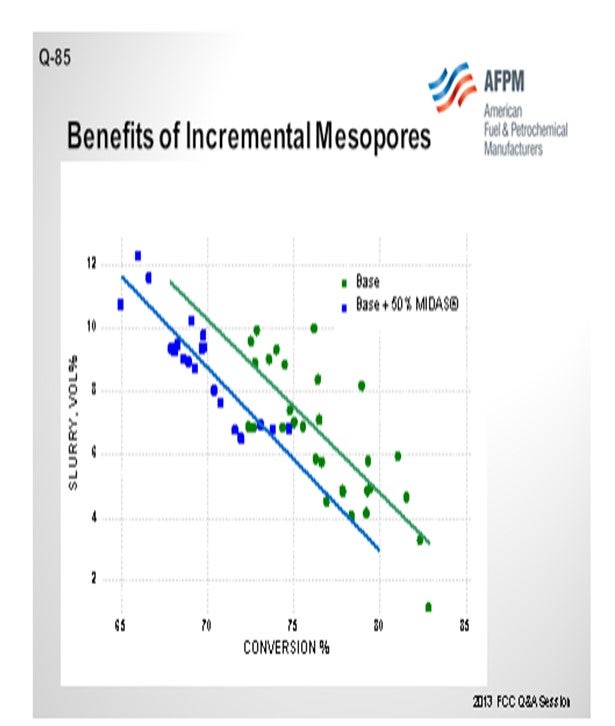
The next slide shows a commercial example of a refinery that was running on a catalyst and implemented 50% of Grace’s MIDAS® catalyst into the blend. MIDAS® is a catalyst for which Grace specifically maximizes the mesoporosity and matrix surface area in the mesopore range. You can see that over a wide range of conversion, the slurry yield was reduced by 2.0% absolute. This is a gas oil operation. Even though this would not be a feed you would consider to be heavy resid, clearly the matrix contribution to the efficiency of the cracking was obvious.
The question also asks about possible disadvantages of mesoporosity. I believe that it might be a disadvantage to have too much mesoporosity in the catalyst when there is a need for high zeolite activity, which is certainly very important, particularly in this Type II cracking where you are going to try and de-alkylate side chains off of naphthenic and aromatic cores. This cracking is very efficiently done by zeolite. If you have a catalyst system that is too biased towards matrix surface area (MSA), you may have a deficit in zeolite activity. So, you certainly want to make sure you have the proper Z/M (zeolite/matrix) ratio on all feeds.
JOE McLEAN (BASF Corporation)
We introduced a catalyst called NaphthaMax® back in about 2000. Really, the only difference between NaphthaMax® and its predecessor-type technologies was that there was a higher porosity version. It has been, by far, our best-selling gas oil catalyst line ever since. We are continuing to refine and introduce newer versions of it. Obviously, I agree with Grace, and certainly with Jeff, that it does make a difference, even in gas oil applications, because it has. We have added over 100 NaphthaMax® users over the past decade or so who could attest to that benefit.
It should not surprise you that we will differ in our opinion a little. I am talking about the zeolite versus the matrix; because with our DMS (dimethyl sulfide) technology, we can put in the mesoporosity. We actually use the external zeolite surface that now gets exposed and available in the larger pores as it functions the way that catalyst is manufactured. So even with the higher porous systems, we are able to get just as high of a zeolite content; and in fact, better zeolite efficiency and activity than what we have with less porous systems generated with the predecessor technologies that we had.
KEN BRUNO (Albemarle Corporation)
Indeed, we agree that mesoporosity is important; but when you talk about diffusion, it extends beyond mesoporosity. In addition to intra-particle diffusion, what is also critical is surface diffusion. To quantify that, Albemarle often talks about the Accessibility Index. Again, mesoporosity is important, but what is more important is the combination of surface and intra-particle diffusion as measured by the Accessibility Index.
PAUL DIDDAMS (Johnson Matthey INTERCAT, Inc.)
Just a quick comment: Do not forget that there are also concentrated additives which contain these kinds of mesoporous cracking sites.
JEFF KOEBEL (Grace Catalysts Technologies)
Effective bottoms cracking involves a three-step bottoms cracking mechanism which was discovered by Zhao1 (Figure 1). The first step is largely dependent on the catalyst matrix and porosity, and that is the effective pre-cracking of large molecules in the feed.2 Since the riser mix zone temperature can be on the order of 1050°F, feed components over 1000°F+ will not readily vaporize. Thus, these molecules need to pre-crack on the catalyst matrix before the catalyst zeolite can further crack them to desired products.
Grace’s FCC feed database contains many examples of units processing VGO and GO that still have a significant portion of the total feed to the unit with a boiling point above 1000°F. In fact, the average 1000°F+ in VGO and GO FCC feed is approximately 10% across our database (Figure 2). Thus, pre-cracking is a real consideration, even in a non-resid operation.
The pore volume of an FCC e-cat (equilibrium catalyst) sample is typically analyzed and reported by each vendor. However, the size distribution of the pores is also a critical component of the effectiveness of the catalyst porosity. Liquid phase diffusion, like one can find in the mix zone of the riser, is typically 2 to 3 orders of magnitude slower than gas phase diffusion, especially if the pores are small (less than 100 Å).4 High mesoporosity in the 100 Å to 600 Å range mentioned in the question is critical for achieving effective pre-cracking of heavy molecules without diffusion limitations. Excess small pores less than 100Å will lead to high coke and gas make, while an abundance of large pores greater than 1000 Å will have ineffective bottoms cracking and perhaps poor attrition characteristics.
Grace’s MIDAS® catalyst family is specifically designed to provide higher matrix surface area (MSA) with an optimal pore size distribution that provides excellent mesoporosity. Grace has many commercial examples of GO and VGO operations that have benefited from the inclusion of some portion of MIDAS® in the fresh catalyst mix. This commercial example shows that inclusion of MIDAS® into a catalyst system for a VGO application improved bottoms conversion by 2% absolute (Figure 3).
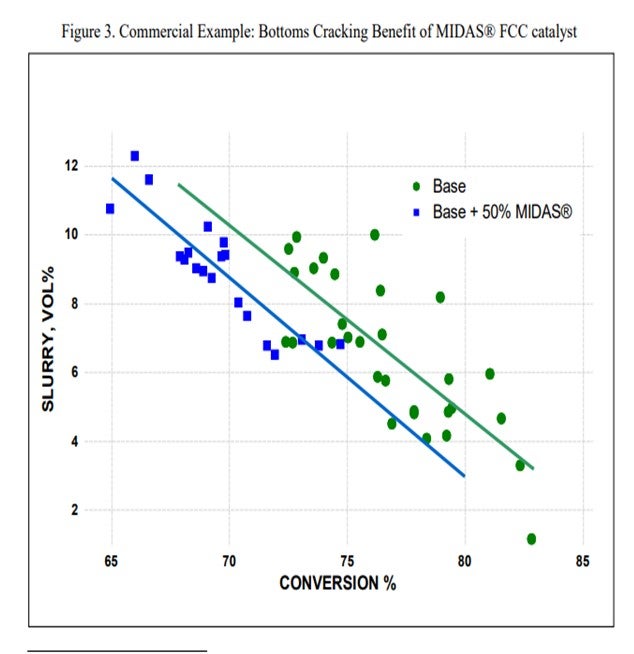
The question also asks about possible disadvantages of mesoporosity. As was mentioned previously, there are multiple steps to effective bottoms cracking. Another step in the conversion of bottoms is dealkylation of side chains from aromatic and naphthenic cores (Figure 1). This is a step that is most efficiently catalyzed by zeolite activity. If a catalyst formulation is too heavily biased towards MSA in an effort to achieve high porosity, it is possible that the formulation may not have adequate ZSA to efficiently drive this type of bottoms conversion. The proper balance between matrix and zeolite is heavily dependent on feed properties and unit yield objectives. Optimization of matrix porosity and zeolite activity is critical to achieve the most effective bottoms cracking, even in light feed applications.
CAREL POUWELS (Albemarle Corporation)
Mesoporosity is typically considered an important feature for enhanced bottoms conversion. This is not only valid for resid applications, but also holds for VGO operations. The effect, however, depends on many aspects, such as the type of VGO, severity of operation, and the base catalyst in use: the more crackable material present in the slurry fraction, the more potential impact by additional mesoporosity. This is particularly valid when the additional mesoporosity is applied within the same catalyst technology. Changing catalyst technology with additional mesoporosity will not necessarily yield the desired effect as the activity, and selectivity of the mesopores between the catalyst technologies can be different. Another aspect that plays a role is catalyst accessibility, which is often confused with catalyst porosity. Poorly designed catalysts can measure high mesoporosity but are not effective as constricted pore openings act as bottlenecks and result in a low accessibility. Thus, it is key to design a catalyst with high mesoporosity and high accessibility.
While mesoporosity (and accessibility) plays an important role in bottoms conversion, it also contributes to the preservation of important primary cracking products such as olefins in the gasoline range. These are important components for high gasoline RON (research octane number) and also precursors for the production of propylene.
Another potential benefit of catalysts with reduced mass transfer limitations in VGO applications becomes evident in units with advanced riser termination devices (RTDs) and short contact times. Under these circumstances, reactants can travel faster to the active sites, and products can egress from the pores before the catalyst is separated from the hydrocarbons in the RTDs, resulting in higher conversion and reduced rate of secondary reactions (everything else constant, compared to a catalyst with inferior pore architecture).
Albemarle offers a wide suite of products for different VGO applications with a varying range in mesoporosity and accessibility, such as AMBER™ for lowest slurry, GO-ULTRA™ for maximum gasoline, ACTION® for superior gasoline octane and alky-feed, and AFX™ for record high propylene.




