Question 61: Extensive use of upstream H2S scavengers in crude oils can cause salt deposition in atmospheric tower overhead systems, as well as on atmospheric tower trays, particularly when trying to operate at lower naphtha endpoints. What adjustments in desalter operation are used to mitigate amine salt deposition?
DION (GE Water & Process Technologies)
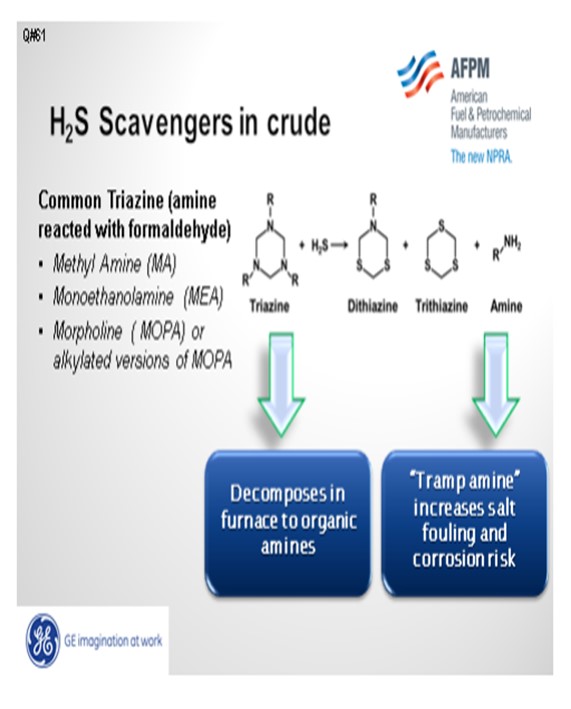
H2S scavengers may cause salt deposition in atmospheric towers. Triazine, the most common scavenger, is made by reacting formaldehyde with an amine, such as MEA (monoethanolamine), and creates an MEA-based triazine; similarly with methylamine-based triazine. One of the byproducts from the reaction of triazine with the hydrogen sulfide is an organic amine. The organic amine, and any unreacted amine from the manufacture of triazine, can increase the amine loading in the overhead, move the salt point, and increase amine salt corrosion risk.
Additionally, the triazine itself can decompose. An MEA-based triazine may decompose to MEA in the crude unit furnace and increase the MEA loading in the overhead. The salt risk is from both a byproduct of triazine reacting with hydrogen sulfide and from the decomposition of triazine in the crude unit furnace. One method to minimize overhead salt issues is to inject an acid upstream of the desalter to achieve an acidic effluent brine pH. An acidic environment will protonate some alkyl amines, thus creating an ionic compound like RNH3+ (protonated alkyl amine) instead of RNH2 (alkyl amine). The ionic species increases its partitioning to the water phase, thereby driving more organic amines into the effluent brine. The partitioning coefficient for those materials is dependent upon the R (alkyl group). In general, the more carbons on the amine, the less likely it will partition into the water phase.
LEE (BP North America)
The primary H2S scavengers used in the industry, as Mike mentioned, are methyl triazine and ethanol triazine. Between the two triazines, we favor the use of methyl triazine due to the lower amine salt point temperatures for the salts that can be formed downstream with the free amine byproducts.
As far as desalter operation impacts from H2S scavenger use, methyl triazine H2S scavengers react to form monomethylamine byproducts. Literature and operating data support that a large portion (greater than 80%) of the amine byproduct will be solubilized into the desalter water if the water is between 6 pH and 8 pH. Lower amine partition percentages will be seen at the higher pH range. The amine partitioning can be improved by lowering pH or even acidifying, as Mike mentioned, but we have not done any of those practices ourselves within BP. As far as dithiazine byproducts, the ethanol dithiazine is reported to be water-soluble, and the methyl dithiazine is reported to be oil-soluble. We do not have any independent quantifiable data to elaborate on these literature reports. High water solubility of the byproducts will support maximizing desalter washwater usage in general.
Reducing the chloride content of the desalter crude will, in turn, suppress the amine salt point of the system. Desalter optimization parameters include maximizing washwater rate, making washwater quality enhancements (particularly pH and ammonia content), conducting mix valve pressure drop optimization, optimizing demulsifier use, optimizing grid voltage settings, and achieving the optimum, usually meaning maximum, operating temperature on the desalters.
VIJAY KRISHNAN (CITGO Petroleum Corporation)
Mike was talking about the use of acids. I would like to know how many people on the panel or in the audience use sulfuric acid to control or reduce pH.
PUI-NANG LIN (Flint Hills Resources, LP)
We have not used sulfuric acid for pH control in desalter application, but we have used it in other applications. We had a very bad experience because this inorganic acid is not buffered, so it is easy to over-inject and get results in a very low pH.
DION (GE Water & Process Technologies)
1,3,5 triazine-based products are commonly used commercially to treat a wide variety of petroleum products throughout the oil production and refining industry to reduce the volatility of hydrogen sulfide (H2S) in these products, making them safer to transport and store. The use of certain chemicals can provide health and safety benefits in shipping petroleum fluids but may also have side effects that are detrimental to refinery operations.
Triazine is a generic term for a small class of chemicals that contain a six-member ring with alternating nitrogen and carbon atoms. Differences between triazine compounds are derived by the atoms bonded to the nitrogen, but not included in the ring structure. Various amines are reacted with formaldehyde to form the triazine. Commercial triazines are comprised of formaldehyde and primary amines including, but not limited to:
• methyl amine (MA),
• monoethanolamine (MEA), and
• morpholine (MOPA) or alkylated versions of MOPA.
The triazine H2S complex that partitions to the oil phase or is carried over in the desalted crude BS&W can decompose in the crude unit furnace. The decomposition products typically contain an amine that adds to the partial pressure in the overhead and, depending on the amine, can form high salting amine chloride deposits.
Impact on Refinery Processes: High triazine levels in crude oil (greater than 1,000 ppm) have been found to negatively impact desalter performance in simulation studies. Triazine that remains in the oil phase will quantitatively decompose to amines at high temperatures in the crude furnace. Depending on the amine used to produce the triazine, a portion of the amine released in the reaction of H2S with triazine will remain in the oil phase and pass through the crude furnace and into the atmospheric tower. Contingent on the level of chlorides present in the desalted crude, amine chloride salts will form and can deposit in process equipment and cause fouling and corrosion. The amine chloride salts are insoluble and heavier than the hydrocarbon fractions and are corrosive to most metals at these temperatures. The type of amine used in the triazine determines where the salt deposition may occur based on the salt point at the specific operating conditions in a specific unit. MEA is known for forming a hydroscopic chloride salts above the gas oil section of the crude tower and is primarily a corrosion problem in the naphtha pump around section and overhead. Should MEA triazine or the sulfur containing reaction product survive the furnace and flash zone, additional concerns include increased sulfur content of the side cut products and fouling. Generally, MA chlorides will deposit in the overhead line or condensers and cause poor distribution, fouling and corrosion in the overhead system. These deposits and resulting corrosion and fouling may negatively impact the performance of the atmospheric tower and result in lost throughput or unplanned outages.
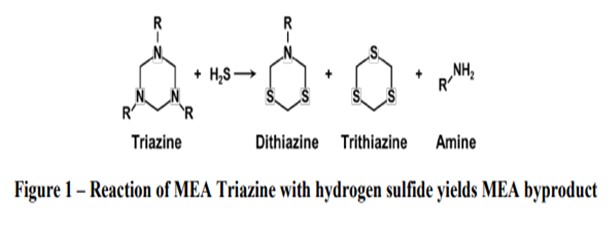
The reaction of triazine and H2S forms heavy sulfur compounds. The ratio of the dithiazine and trithiane that are formed depends on the relative ratio of reactants present. In most cases, dithiazine is the predominant species. Trithiane is more difficult to form, but it is more likely to form in the presence of excess H2S and/or low pH conditions. Both of these molecules can decompose in the crude furnace and distill into the refined product cuts. Dithiazine is hydrocarbon soluble and most remains in the oil phase. It distills at ~300°C (572°F) and may distill into middle distillate cuts. Some has been found to decompose to form CS2 (carbon disulfide) and free amine. Trithiane is generally insoluble in water and hydrocarbon. These solids that are dispersed in oil can sublime at >250°C (482°F).
1. Effects on Wastewater Treatment Plant (WWTP): All of the amines from triazine and reaction products will eventually flow to the WWTP. The amount of nitrogen will be the same regardless of amine since the stoichiometry of the triazine reaction is dependent on the number of Formaldehyde molecules that are consumed and not the molecular weight of the amine. These amines are readily consumed in the wastewater plant but may result in sudden microorganism growth and reproduction, followed by depletion of the oxygen supply. If the oxygen supply cannot be replenished, the microorganisms die from lack of oxygen and must be repopulated. The impact to the WWTP can be minimized by either limiting the blend ratio of triazine-treated crude oil or inoculating the WWTP with Nitrification organisms.
2. Carbon Disulfide: Field Experience demonstrates a 10-time increase in CS2 content in naphtha produced from triazine-treated crude. This is expected from any triazine since the CS2 is formed when the dithiazine cleaves, as shown below
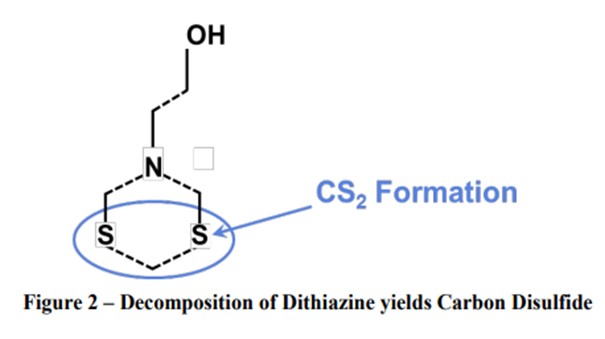
Small amounts of carbon disulfide and dimethyl sulfides may be formed as a result of thermal decomposition or reactions with other chemical species or the reaction product of MEA triazine and H2S. This does not create a problem for most refiners but could be a problem in petrochemical plant reactions because it is a catalyst poison. There are very few refineries in the world that send naphtha directly to petrochemical processes and may be affected.
The impacts of triazine-treated crude can be mitigated in several ways. The crude buyer must request information on treatment levels if they suspect that crude oil cargoes have been treated for H2S. Once this information is obtained, the crude blend rate can be controlled to limit the amine content in the crude.
Alternately, an acid can be added to the crude and/or desalter washwater to maintain an acidic effluent brine pH. The reduced pH protonates amines and creates an ionic compound that is more soluble in the bulk water phase, thereby extracting amines from the crude into the effluent brine. The types of amines and extraction efficiency will be dependent on the type of amine involved. In general, the more carbons there are on the amine molecule, the less efficient the extraction. For example, ammonia will be easier to extract than MEA and MEA easier to extract than DMEA.
If possible, the best solution is removing the potential processing hazard completely. The refiner may request that the crude producer or broker use a non-amine based H2S scavenger such as a select GE Water & Process Technologies’ non-amine ProSweet* product to completely eliminate the potential impacts of H2S scavenger treatments across the entire refinery.
RANDY RECHTIEN (Baker Hughes)
When amine-based H2S scavengers are present in crude oils, desalter operating conditions affect the extent to which amines partition to the desalted crude. The key operating parameter is desalter water pH. At higher pHs, higher levels of amines partition to the crude and increase the risk for amine-HCl salt formation in the atmospheric tower and overhead system. The application of desalter acidification additives, such as Baker Hughes EXCALIBUR™ program, can successfully decrease amine levels in desalted crude. In conjunction with an EXCALIBUR™ program, the potential for amine/HCl salt formation can be determined using Baker Hughes TOPGUARD™ Corrosion Risk Monitor.
BASHAM (Marathon Petroleum Corporation)
Control of the desalter water pH is key. Amines will partition between oil and water depending on pH. Amines will go with the water at lower pHs, and conversely stay with the oil at higher pHs. This can lead to cycling up of amines in the system.
DENNIS HAYNES (Nalco Energy Services)
Elevation of washwater flow rates, injection strategies (moving towards a more dominantly primary versus secondary injection where possible), not using desalter washwater with an elevated amine loading, and pH control of desalter washwater are all strategies that will assist to certain degrees.
SAM LORDO (Nalco Energy Services)
Acidification of the desalter washwater, typically using organic acids, can be done and achieve some success in extracting amines from the crude oil. This does have its downside, though, and has to be monitored closely or one could end up with additional acid loading on the overhead circuit. This additional acid loading could result in additional neutralizers being used, which could then also raise the salting temperature of the chloride salts. A thorough review of the system to be treated should be conducted so that the proper acid is chosen and the side effects are minimized.
Question 62: What are causes of foaming in crude pre-flash drums and towers, and what options are available to mitigate foaming?
SHELTON (KBC Advanced Technologies, Inc.)
Surfactants cause foaming. Mike will discuss surfactants and amines that should not be in the crude. Sodium naphthenate is a common surfactant produced by the reaction of caustic injected at the desalter effluent and naphthenic hydrocarbons.
A simple solution is to move the injection downstream of the pre-flash or pre-fractionator to the bottom pumps. If the injection point is at the desalter effluent, solids and corrosion products can cause foaming.
Improving desalter solids removal will mitigate foaming. Precipitated asphaltenes that frequently occur with bitumens and asphaltic crudes can also cause foaming, so we would evaluate crude compatibility in that case.

The question includes pre-flash drums and towers, which I assume is a pre-fractionator. These applications are quite different in design and operation. In our pre-fractionator designs, we consider C factors, internals, tray design, and tower loadings.
We have used pre-flash drums in our latest grassroots designs because the hot trains have been so efficient that crude heater inlet temperatures are 600ºF to 610ºF. A flash drum removes water and requires lower pressure to suppress vaporization at the end of the hot train. The flash drum design avoids elevated pressures in the hot train and 900-pound flanges. Obviously, we specify vertical versus horizontal. We consider height versus diameter and liquid superficial velocity versus vapor velocity to optimize the ratio. We also consider disengaging height and the feed distributor inlet design. Of course, temperature and pressure have a major impact.

The pre-flash drum performs two functions: flashing water and suppressing vaporization. Many pre-flash drums are operated to remove light hydrocarbons. However, water causes vaporization, and operating pressure and temperature determine the vapor rate and composition. It is important to model the optimum pressure. Operate at the pressure required to remove water and not generate excessive hydrocarbon vapor load, which can result in carryover of bottoms.
In our designs, flashed vapors are sent to the flash zone. Designs where the flashed vapors are introduced in higher sections of the column can create problems. For any design, in the event of a foamover, temporarily increase pressure. With the flash drum, increase pressure until there is no vaporization. That will stop the foamover. It is important to have a pressure controller on that vapor line to the flash drum.

Pre-fractionator foaming is less likely because it is a refluxed column with an overhead product. The trays mitigate foaming, and the liquid loading should tend to knock down the foam. Again, in the event of a foamover, you could temporarily increase pressure. This may not be obvious, but we try to design for higher temperatures to reduce surface tension, which also mitigates foaming. In a new design, the pre-flash drum operating temperature is determined by the location in the hot train. Finally, improving desalter operation will mitigate foaming in the downstream columns.

BASHAM (Marathon Petroleum Corporation)
I want to reinforce some of Al’s points here. As he already mentioned, pre-flash tower or vessel foaming is a function of crude type salt or water carryover, temperature, and caustic addition. You are always going to have foaming occurring in a pre-flash drum or tower. The key here is to manage the foam and keep it in the tower. You must have sufficient vessel height and diameter necessary to disengage the foam. As Al also mentioned, the liquid superficial velocity is the key design parameter. It is important to keep in mind that the smaller the diameter of the vessel, the larger the foam height; so in narrow vessels, the liquid superficial velocity will need to be low in order to keep the foam height low. It is possible to add silicone-based antifoam to the pre-flash drum or tower, but consideration needs to be given to the downstream, gasoline, and distillate hydrotreater reactor catalyst.

DION (GE Water & Process Technologies)
Al and Kevin covered operational and mechanical issues regarding foam. Part of the question asked about the causes of foaming. There are surfactants in crude oil. Surfactants can be any organic molecule that has an atom that is not carbon or hydrogen, such as organic acids, organic amines, mercaptans, and other molecules with a polar group associated with them.

RUSSELL STRONG (Champion Technologies)
I have heard several comments that silicone antifoams in crude are problematic. There have been recent events offshore in the Gulf where so much antifoam was being used upstream that it was actually poisoning hydrotreater catalyst in the refinery from the upstream application. Other causes of silicone contamination can come from the crude while trying to control foaming in a flash drum or in a crude tower. To control those, silicone antifoams are sometimes used with occasional success. Several years ago, at a refinery down in the Houston, Texas area, I encountered severe foaming in a crude tower that would not go away. Standard silicone antifoams did nothing to solve the problem, but a fluorosilicone antifoam worked well. It was far more efficient and actually worked where the polysiloxane was deficient. It also offered less risk of downstream silicon contamination. So, keep this in mind as an option if you have crude unit foaming.
STEVEN FISCHER (Delek Refining)
At a previous refinery, we reintroduced the vapors to the flash zone with the result being quench to the flash drum that resulted in poor cutpoints. When we introduced the flash vapors from the flash drum to the flash zone, we saw that that the flash drum had actually acted like a quench, which could result in a poor cutpoint at the bottom of the crude tower.
SHELTON (KBC Advanced Technologies, Inc.)
Simulations do not indicate flash zone quenching if, as previously mentioned, the flash drum operating pressure is optimized to flash-only water. We have evaluated the flow schemes in models with the two streams mixed outside of the column and combined in the flash zone, but we get the same overflash.
STEVEN FISCHER (Delek Refining)
That was our assumption when we designed it that way, but our performance did not show that result. Our performance improved when we introduced it higher up.
ANDREW SLOLEY (CH2MHILL)
Addressing that last comment, I think what you are seeing there, when you see the poor performance, is the mixing of transfer line liquid with the vapor coming in, which is an issue with the equipment and not having the vapor segregated from the transfer line.
SHELTON (KBC Advanced Technologies, Inc.)
Our designs do have a separate flash drum vapor nozzle in the flash zone. It is important to have a pressure controller on the flashed vapor line, so the drum is not riding on the lower flash zone pressure. I do not know if that is your case or not. Do you have pressure control on the pre-flash drum? If not, a large pressure drop will produce a very high vapor rate, and then hydrocarbons will be flashed. In that case, there could be some quenching. We try to just flash the water and no hydrocarbons. When you think about it, if there were substantial light hydrocarbons, the desalter would overpressure. So, there are not a lot of light hydrocarbons in the crude because the only difference in the flash drum versus desalter operation is the desalter pressure, which is also low compared to the elevated hot train pressure.
STEVEN FISCHER (Delek Refining) We had some light hydrocarbons going overhead in addition to water.
SHELTON (KBC Advanced Technologies, Inc.)
There may also be recycle streams quenching the flash zone.
ROBERTSON (AFPM) Al, could you comment on the superficial velocity?
SHELTON (KBC Advanced Technologies, Inc.)
Liquid superficial velocity is a function of the vessel height versus diameter and design of the drum, which differs for vertical versus horizontal vessels. It is specific to each design and not a variable for an existing drum. Pressure is the important operating variable. If there is no pressure controller on the vapor from the flash drum, then that deficiency can be remedied online because there is usually a block valve at the column. In that case, the back pressure controller can be installed online.
VILAS LONAKADI (Foster Wheeler USA Corporation)
Is there any experience with the use of any internals in the pre-flash drums?
SHELTON (KBC Advanced Technologies, Inc.)
There are several types of feed distributors, including vortex tube clusters (VTC) and tangential nozzles. There are many effective feed distributors that will improve disengaging.
VILAS LONAKADI (Foster Wheeler USA Corporation)
Not about just the feed entry, but in the drum itself.
SHELTON (KBC Advanced Technologies, Inc.)
We do not recommend demisters on vapor outlets, and flash drums do not typically have any internals.
VILAS LONAKADI (Foster Wheeler USA Corporation)
Some vendors offer vortex tube clusters. I want to know if anyone has used them.
SHELTON (KBC Advanced Technologies, Inc.)
Yes, we mentioned vortex tube clusters (VTC), which have been used successfully in drums that operate at high velocities. We have also seen VTC distributors used for revamps to increase throughput at higher drum velocities. They have been very effective.
VILAS LONAKADI (Foster Wheeler USA Corporation)
Did it reduce foaming?
SHELTON (KBC Advanced Technologies, Inc.) Yes, VTC distributors have been used to solve foaming problems for existing vessels.
SHELTON (KBC Advanced Technologies, Inc.)
Foaming in flash drums and pre-fractionators is often caused by crude contaminants. Inorganic fines (sand, corrosion products, etc.), precipitated asphaltenes and sodium naphthenates formed from the reaction of caustic and naphthenic hydrocarbons have been identified as precursors. If caustic is injected at the desalter effluent, a simple solution is to move the caustic injection downstream of the flash drum to the pre-flash bottoms or hot train pumps.
The immediate solution to a foaming problem is to increase pressure to decrease vaporization. In a prefractionator, in addition to increasing pressure, higher reflux or wash rates will tend to knock down the foam front. Increasing temperature will reduce surface tension and mitigate foaming. Long term solutions include improving desalter operation (particularly solids removal) and improved selection of treating chemicals for the preheat train and desalters.
The design and operation of pre-flash drums and refluxed pre-fractionator columns are different. Vessel design (vertical versus horizontal) and disengaging height affect foaming. KBC design guidelines for pre-flash drums include height versus diameter, liquid superficial velocity versus vapor velocity, disengaging parameters, feed distributors and pressure. For any design, increasing operating pressure will reduce foaming.
Pre-flash drums are located in the hot crude train downstream of the desalters to flash water and suppress vaporization at the end of the hot train. Flash drum vapors on pressure control are routed to the crude column flash zone. Flash drum pressure sets the vapor rate and composition. Simulations show that water causes vaporization in heat exchanger services at the end of the hot train, not light hydrocarbons. Very light hydrocarbons would overpressure the desalters, if present. Simulations will determine the flash drum pressure required to remove dissolved water from the desalter effluent. The flash drum should be operated at the pressure required to remove water and no lower to reduce carryover of flashed crude. In the event of a foamover, the foam can be broken by temporarily increasing drum pressure to reduce vaporization. Good desalter operation with no water carryover to the flash drum will minimize foaming. Desalters should be operated with less than 0.5% BS&W in the effluent. Prefractionators are typically refluxed distillation columns with an overhead product such as light naphtha and may also have sidecuts. Foaming is less prevalent in a refluxed column. In the event of a foamover the foam front can be broken by first increasing reflux rate and if necessary, temporarily increasing overhead pressure.
BASHAM (Marathon Petroleum Corporation)
Foaming is always present in pre-flash drums and towers. It can be a function of several parameters including crude type, desalter performance (water carryover), drum or tower temperature, and caustic addition. Depending on its feed location in the atmospheric crude tower, pre-flash drum vapor can cause black distillate, black atmospheric gas oil, and increased atmospheric tower bottoms if the foam contains flashed crude. Similarly, in pre-flash towers foam with entrained flashed crude can cause black naphtha. The key to managing foam is keeping it in the pre-flash drum or tower.
A properly designed vessel (drum or tower) will allow sufficient height to disengage the vapor from the liquid. The most important design parameter is the superficial velocity of the flashed crude. The foam height is directly proportional to the liquid superficial velocity. The liquid superficial velocity must be sufficiently low enough to keep the foam height below the vapor outlet of the drum or tower. The foam height is also a function of the tower or drum diameter (cross-sectional area.): the smaller the diameter, the larger the foam height. This means that foaming will be a bigger concern in narrow vessels, so the liquid superficial velocity will need to be low in order to keep the foam height low.
It is possible to add silicone-based antifoam to the pre-flash drum or tower, but consideration must be given to downstream gasoline and distillate hydrotreater catalyst silicon loading.
LEE (BP Products North America)
A potential cause is water carryover out of the desalter that is vaporized in the flash drum. If there is water carryover and high shear stresses associated with a letdown valve with high pressure drop, this situation can generate small droplets which would contribute to foam generation. Foaming is often associated with high vapor rates, so a crude with a significant amount of vaporization at the flash drum conditions may have high potential for foaming. Antifoam use, and additional enhanced separations hardware, such as vortex cluster internals, can be considered.
DION (GE Water & Process Technologies)
Any organic molecules with atoms other than hydrogen or carbon are potential surfactants. Examples of such molecules are; alkyl phenols, organic amines, organic acids, and mercaptans. Foaming can be mitigated through the use of a defoamer or antifoam chemistry.Defoamers function by reducing the interfacial surface tension and viscosity. Antifoams function by modifying the interfacial surface elasticity. Most products commercially available from specialty chemical suppliers, such as GE Water & Process Technologies, function in both manners due to the behavior of their surfactant structure. The most effective defoamers in hydrocarbon environments are typically silicone based. If silicone poisoning is a concern, non-silicone-based defoamers, such as glycolic materials, are available.
BRUCE WRIGHT (Baker Hughes) Pre-flash tower foaming is most often caused by high solids loading coupled with high gas flows. Foam control with Baker Hughes Si-based antifoams has proven to be effective.
DENNIS HAYNES (Nalco Energy Services)
Crude viscosity, hydrocarbon polarity, solids content, caustic use, and vapor disengaging in flash sections and tower bottoms are discussed as causes for foaming. Antifoams have been around for quite a while that may be utilized in this area; however, the first step in corrective action is to determine that it is actually a stabilized foam layer and not tower flooding. There are instances where pre-flash towers are operated above design or have had some internal damage that causes flooding which is mistaken for foaming.
ANDREW SLOLEY (CH2M HILL)
One major cause of foam formation in these units is surface-active agents stabilizing the foam film on the liquid-vapor interface. Some of these agents are inherent components of specific crudes. However, many of them have been added to crude as well stimulation, drag-reducing, anticorrosion, or hydrogen sulfide scavenging additives. With continued production of heavier crudes and more aggressive well stimulation operations, foaming problems should be expected to get worse.
Solutions to foam formation include; antifoaming additives; foam-breaking inertial separators; and modifying operating conditions.
Silicone-based antifoaming additives can be effectively used. Their downside is that they vaporize and end up in the lighter products, particularly naphtha. This puts the antifoam into the downstream naphtha hydrotreater feed. Few hydrotreaters can tolerate this. Antifoams are rarely used.
Foam-breaking inertial separators have been used in a number of plants. They are derived from equipment design for oil production operations. In the oil fields they are proven technology. Experience in refineries, while limited, has been mostly successful. For certain plants and feeds they may be a choice worth serious consideration.
The most common method of avoiding foam-created problems has been to modify the plant operating conditions. This may include changes in feed rate, pressure, or temperature. Feed rate reduction increases effective residence time in equipment. It also reduces total vapor rate formation. While expensive, some plants are constrained to do this. Increasing pressure reduces vapor formation and increases vapor density. Both reduce the volume of vapor. Increasing operating pressure reduces foam problems. Temperature changes are more complex. Higher temperatures (at the same pressure) create more vapor volume, they also decrease liquid viscosity. These are competing changes. More vapor volume increases foam make. Lower viscosity speeds foam decay. In a plant with a foam problem, small temperature changes, in either direction, may help solve the problem. Experience has shown an operating temperature change as little as 10°F may change the vapor volume, or the viscosity, enough allow the flash drum or tower work, or be catastrophically worse.
Proper pre-flash installation includes balancing many factors including equipment size, expected operating conditions, and how to connect the pre-flash system to the existing unit. Revamps to add, or improve, pre-flash drums or towers need to be carefully evaluated.
Question 63: Crude and vacuum tower off gas production from bitumen crudes can be quite variable depending on feedstock quality. Please comment on observed off gas production when processing bitumen crudes.
LEE (BP North America)
Our response to this particular question is based on the presumption that bitumen crudes include the conventional Canadian heavy supply of crudes such as Lloydminster and Cold Lake. There has not been much Canadian tar sands bitumen actually processed within BP, either as a synbit (bitumen diluted with synthetic crude) or a dilbit (bitumen diluted with condensate) as of yet. Some of this new bitumen supply includes supplies such as Christina Lake, Sunrise, and Firebag bitumen. The vacuum resid cut has a very high asphaltenes content, and we expect to see a significant cracked gas production rate. The cracked gas production will vary largely with the vacuum furnace thermal severity, mainly film temperature, residence time, and feed quality. We do not see any particular notable cracked gas production from many of our crude furnace operations.
There are other factors, besides those cited, that can also contribute to gas production. If the bottom surge volume in the vacuum tower has a long residence time, say above three minutes, and it is also unquenched, then gas can be generated by cracking in this zone. We typically like to quench this zone to less than 680°F or so. Another mitigating parameter for gas cracking is the use of coil steam to increase furnace tube velocity and minimize residence time and film temperature. Most of our high severity vacuum furnaces utilize coil steam.
We generally correlate the furnace feed quality to API gravity, but the feedstock factors need to be applied based on experience with the supply source. Generally, heavier, more asphaltenic crudes will produce more off gas at a given furnace temperature. Feedstock factors are important as we know that there are more reactive and less reactive asphaltenes content present. There are also not a lot of correlated or quantified experiences with gas production rates. We have found that our conventional Canadian heavy oil supply, for example, produces roughly one and a half times the gas make predicted by a base cracked gas correlation we use. We expect the synbit or dilbit supply to produce even higher cracked gas production rates. We also recognize that once a feedstock produces a significantly higher off gas production rate than this, it will likely become uneconomical to run higher severity furnace operation – that is, higher cutpoint operation – due to the costs associated with attendant coking, fouling, and other cycle life maintenance expenses.
Question 64: Based on your experience, what are causes of fouling in the diesel/distillate draws of crude, vacuum, and coker fractionation towers? Does this migrate to downstream diesel hydrotreating units? What mitigation strategies are being employed to overcome these issues?
LEE (BP North America)
We have been concerned with potential phosphorous fouling on units processing Canadian heavy crude oil. We are aware that some refiners have experienced and reported on significant phosphorus fouling issues in the light diesel or jet draw section of the crude tower, as well as in the crude furnace. While we are concerned with the issue and monitor for it, we have not had any particular callout issues with CDU phosphorus fouling. In our view, the fouling of interest in the diesel draw section is differentiated from the amine or ammonia salt fouling that we typically see in the cooler top section of the tower. These particular salts condense at colder temperatures about less than 300°F, and we have not typically seen issues with these salts in the diesel section except for a particular example that I will describe shortly.
We have converted from conventional moving valve trays to fixed valve trays in the diesel section of a crude tower to help mitigate fouling. However, we have not yet definitively demonstrated the benefit of the modification as it is difficult to achieve site-by-site comparative conditions for doing such. This is a preemptive type of tower internals modification.
Many of our refineries are particularly concerned with any downstream unit impacts. We take phosphorus samples in our CDU products and closely monitor if issues are suspected or if there is a phosphorous-bearing crude scheduled or suspected to be in the crude supply. In the table below, you can see examples of some suspect Canadian crudes. The phosphorous contents are shown for these crudes. We monitor unit health and operations closely on our CDU blends that are in the 2 ppm to 3 ppm phosphorous content range.

Let me add that one example I said I would give you. In this case, we did get some diesel section salt fouling. This one happened to be on a coker diesel draw tray. We saw nozzle corrosion on this unit that was due to chloride and salt pitting corrosion and which was the result of both wet reflux and lower tower temperature profiles during drum swings. This particular coker has had issues with wider-than-typical temperature swings during the coke drum switches and a relative deficiency of warmup capacity, both of which contributed to the lower temperature profile seen on the coker fractionator. So, chlorides in this coker naphtha and diesel products did find their way to downstream units in this particular refiner.

BASHAM (Marathon Petroleum Corporation)
We experienced a similar situation as Howard just mentioned. At our Robinson Refinery crude unit, we have seen phosphorous fouling over the kerosene section for about three to five trays. We also had floating valves that we replaced with a fixed valve design to help out with that fouling as well. In a coker fractionator tower, it is important that, as Howard said, you do not let the overhead temperature get too low; because when you force the colder temperatures down in the tower, you can see the salt formation in the distillate section.
DION (GE Water & Process Technologies)
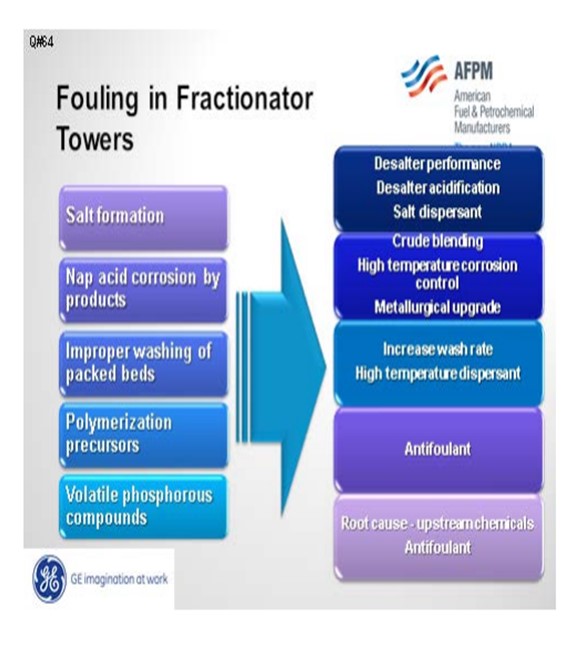
Fouling in the distillate trays and other locations can be divided into five different categories: salt, corrosion byproducts, coke, polymers, and volatile phosphorous compounds. Amine salt fouling can be mitigated with improved desalter performance, removal of tramp amines, and the use of a salt dispersant. Acidifying the desalter will partition a greater number of amines into the effluent brine.
If you do not have the proper metallurgy, corrosion byproducts can foul downstream units. This can be addressed with metallurgical upgrades and high temperature corrosion control programs, as well as blend control to minimize the TAN (total acid number) and NAN (naphthenic acid number) of the crude blend.
Improper washing of packed beds can contribute to fouling. This condition is usually seen in vacuum towers that have high surface area and very high molecular weight heavy hydrocarbon molecules that may precipitate, dehydrogenate, and create coke. This fouling can be addressed operationally with wash rates and the use of a high temperature dispersant to move coke particles.
Bitumen streams, such as dilbit from Canada and DCO (diluted crude oil) from Venezuela, may have the potential to induce polymerization. These crudes are typically bitumen with a diluent added to reduce viscosity. If these diluents come from cracking processes, there is the potential that they may contain olefins which could encourage fouling from polymerization products.
Howard addressed volatile phosphorus. These are most likely from phosphorus-based oil field chemical additives.
AHMAD AL-JEMAZ (Kuwait National Petroleum Company)
Again, I want to ask about mitigating fouling in the coker fractionator. Also, how effective is the introduction of online antifoulants to clear the fouling material created from corrosion products or coke fines and salts? Is there any record of successful implementation of antifoulants to clear the fractionators?
DION (GE Water & Process Technologies)
It is easier to prevent fouling from occurring than to remediate after the fact. An example is asphaltene precipitation. It is easier to prevent asphaltene molecules from agglomerating to a size where they precipitate than it is to redisperse them once they are agglomerated. Sloughing the tower to wash fouling material out of the system is usually a last resort. A better approach would be to examine methods to minimize coke entrainment into the tower including: an effective coker defoamer, defoamer to diluent ratio, coker overhead corrosion inhibitors, upward vapor velocities in coke drum vapor line, and an antifoulant in the gas oil or other reflux.
CELSO PAJARO (Sulzer Chemtech USA, Inc.)
For our delayed coker main fractionator, we have successfully used antifouling trays (VG AF™). They have several advantageous characteristics to mitigate fouling including fixed valves, push valves, fouling-resistant downcomer designs, and special outlet weirs. These features generally resist fouling and/or serve to move sediment across the trays to minimize accumulation of these fouling components.
LEE (BP North America)
I have one other comment on the first question: the use of antifoulants. Maybe the vendors know more about this, but I think there is some literature or some publications where folks have successively used iron dispersant to move around the iron scale. We considered that for the salted-up tower we had; but to us, it felt very much like a last resort because if we moved those solids to a draw nozzle or somewhere else, we would be shutting down the unit. So, we considered it, but we thought it was too risky to try.
BASHAM (Marathon Petroleum Corporation)
At Marathon Petroleum Company, the only issue we have had is the phosphorus fouling in our Robinson refinery crude tower, which only occurred over three to five trays in the kerosene section. We have reason to believe that phosphorus will migrate to both the kerosene and diesel sections, thus affecting the diesel hydrotreater. However, only the volatile phosphorus is believed to result in tower fouling. We replaced the trays (floating valve) with an antifouling fixed valve design.
DION (GE Water & Process Technologies)
There are five primary ways that foulants can be generated and deposited in the diesel/distillate sections:
Salt Formation: With more focus on diesel maximization, more tower operations have been changed to run at lower overhead temperatures. The shift in tower operation coupled with tramp amines either in the crudes or recycled via other streams can cause salting in the tower. The salts will foul the tower and since they are very corrosive, will end up generating corrosion byproduct which will also contribute to the fouling.
Naphthenic Acid Corrosion: Naphthenic acids can distill and concentrate in these cuts. If the temperature is high enough and the metallurgy has not been properly upgraded (SS 316L and SS 317L), corrosion will occur. Depending on the severity of the corrosion, the fouling rate from corrosion byproducts can be quite significant.
Improper Washing of Packed Section: This typically happens more in vacuum towers where packed beds are more common. There is more surface area for compounds such as asphaltenes and other heavy molecular weight material to deposit and dehydrogenate forming coke. Some of the coke fines will end up going with the draw and can foul the downstream exchangers.
Polymerization Precursors: With the increased availability of dilbit, synbit and diluted crude oil, there is an increased risk the diluent used for these synthetic crude or bitumen products are from cracking processes. If the diluent is from a cracking process and either not hydrotreated or the hydrotreater is not completely efficient, polymerization precursors can be present.
Volatile Phosphorous Compounds: With the increased availability of unconventional crudes, fouling from volatile phosphorous compounds is becoming more prevalent.
All of these foulants and foulant precursors have the potential to impact downstream units.
Mitigation Strategies
1. Salt formation:
a. Desalter performance improvement (chloride reduction)
b. Desalter acidification to reduce tramp amines
c. Salt dispersants
2. Naphthenic corrosion:
a. Crude blending to reduce naphthenic acid content
b. High temperature corrosion control program (chemical treatment)
c. Metallurgical upgrades
3. Improper wash:
a. Increase wash rate.
b. Apply high temperature dispersant.
4. Polymerization precursors
a. Apply antifoulant chemistry to minimize polymerization.
5. Volatile phosphorous
a. Perform root cause analysis of upstream chemical additives to minimize or eliminate the risk.
b. Apply of antifoulant chemistry, such as dispersant, to minimize fouling due to phosphorous compounds.
BRUCE WRIGHT (Baker Hughes)
Salt deposition is the most common. Both ammonium and amine chloride salts have been identified. The salts most likely will not migrate downstream, but can lead to additional iron sulfide formation, via underdeposit corrosion, which has been seen to migrate downstream. The deposition of the salts is a function of temperature and concentration. Identification of the operating window in which salts will deposit can be determined by use of the Baker Hughes Ionic Model. The Ionic Model can also be used to select the most appropriate neutralizer to reduce salt deposition for the operating window. Baker Hughes salt dispersant additives have also been successfully employed. iron sulfide fouling is also not uncommon. Effective mitigation has been achieved with Baker Hughes corrosion inhibitors and/or iron sulfide dispersant additives. Organic phosphorus compound deposition has also been experience. Both mitigation and online cleanup have been achieved with Baker Hughes additive application.
DENNIS HAYNES (Nalco Energy Services)
Diesel/distillate draw fouling in distillation columns is distinct from the problems reported in the literature for naphtha section fouling. Fouling that has been seen at this area lower in the tower, in certain cases, has been related to some specific chemicals used in the production of crude oil. The Canadian Crude Quality Technical Association has done projects in this area over past years. Where the fouling has become apparent in columns, dispersant type chemistries have been successful in reducing material buildup.
SAM LORDO (Nalco Energy Services)
In this part of the tower, amine/ammonia salting is the primary fouling encountered. The following has been done to minimize this type of following:
• Raise tower overhead temperatures,
• Optimize desalting operation to minimize chloride and/or amine traffic in the tower, and/or,
• Use salt dispersant chemistries.
Question 65: Our vacuum column wash bed has lasted seven years in service and now needs to be replaced due to excessive coking and pressure drop. What is the typical life expectancy of the wash grid and packing? What is the panel's experience for the use of wash oil to the vacuum column wash section bed in gpm/ft2 (gallons per minute per square foot) with structured packing and/or grids in the bed? What is the recommended maximum slop wax draw temperature? Should a limit be set on this temperatu
SHELTON (KBC Advanced Technologies, Inc.)
This topic was discussed extensively in the previous Q&A, so my response will focus on wash bed replacement after seven years due to excessive coking and pressure drop. The typical life expectancy of a wash bed has to be qualified. It is dependent on cutpoint, design of the column, internals, operation, and severity.
A wet operation with stripping steam requires lower coil outlet, severity, and flash zone temperature to achieve the same cutpoint compared to a dry operation. I will not address lube oil wash beds, which last considerably longer than a fuels operation. Obviously, a bed with 100% grid lasts longer than a deep-cut design with structured packing.
Since the run length is seven years, it is assumed this is combination packing over grid and a deep-cut design. For a heavy crude operation, six-year runs are typical. If it is a severe operation, then even with a well-designed wash zone with good overflash and adequate slop oil there are still some sections of the packing that have to be replaced during turnaround. With some asphaltic crudes, refiners are satisfied if they make a run without any incidents. So I would say that a seven-year service life is better than average in a severe operation.

Regarding the specific wash oil flux rate, we try to avoid rules of thumb. Every operation is different and has to be modeled. In this kind of severe operation, the depletion ratio could vary from 4:1 to 6:1 and is a function of bed height and wash rate conditions. In a severe operation or deep-cut operation, we typically see 80% of the wash oil vaporized in the beds. KBC targets the overflash or the slop oil at the bottom of the bed, and the required wash oil rate at the top of the bed to produce that overflash target is variable.
It was interesting when we were talking amongst the panel. I would have thought this would have been the standard. Recent Q&A answers quote flux rates, but it was surprising that standard conditions or flowing conditions were never specified. At the low end, the cold versus hot flux rate varies by 35%.
Typical overflash flux rates range from 0.1 gpm/ft2 to 0.2 gpm/ft2 at 60°F, and 0.1 gpm/ft2 at 60°F is about 0.15 gpm/ft2 at operating temperature. In our designs, we target 0.20 gpm/ft2 overflash at 60°F at the bottom of the bed. The wash oil rate at the top of the bed is whatever is required to attain 0.20 gpm/ft2 at 60°F overflash at the bottom of the bed. Wash oil rate is a function of flash zone temperature and pressure. Hopefully, if you work on an operating unit, it has a slop oil meter. Then the wash oil rate should be adjusted to maintain the target overflash at the bottom of the bed. This is how we model an existing operation that does not have a slop oil meter. Frequently, the wash oil to the top of the bed is measured but not the slop oil or overflash.

For an optimization or a grassroots design, we develop a fully-integrated model of the vacuum heater with our VIS-SIMTM model, including the transfer line and the wash zone in the column. The overflash in the column model and the heater outlet temperature are specified.
Petro-SIMTM accurately predicts the depletion ratio based on theoretical stages, and the wash oil rate at the top of the bed is variable. The answer to the target wash oil rate is whatever is required to produce the desired overflash at the bottom of the bed and which will ensure that the wash bed will operate trouble-free for a run-length.

Discussions about wash oil should include inlet distributors because this is critical. In our grassroots designs, we specify spray distributors with 200% to 300% coverage; so, in any one spray pattern, even if one or two spray nozzles fail, there is still coverage in that zone. Some vendors use tough distributors, but we have designed 47-foot ID (inside diameter) vacuum columns. It is difficult to install a level gravity flow distribution system in a very large diameter column. Furthermore, in a deep-cut severe operation, we would be concerned about coke solids plugging the gravity troughs. With any distribution system, proper metallurgy is very important with a good filtration system to remove solids from the wash oil.
The final question was about the recommended maximum slop wax draw temperature. The slop oil temperature should be cooler than the flash zone, which confirms that the wash oil has penetrated the bed and will not coke. As far as a maximum temperature, Kevin thought this might have to do with pump seals.

BASHAM (Marathon Petroleum Corporation)
In our experience, if the wash bed temperatures get up over 770°F or so, the coking tendency will go up. We have had beds in mild service that have lasted over 10 years. We have had beds in severe services that have only lasted a couple of years. As Al was saying, it really depends on the severity of the service. In regard to the maximum slop wax draw temperature, one of the considerations needs to be the slop wax pump seal integrity. We try to keep the slop wax pump temperature below 700°F. We do that with vacuum tower bottoms quench that is cooled and put back to the pump suction.

CLAY MARBRY (Roddey Engineering)
Does anyone on the panel or in the audience have any experience with recycling spent wash oil from the vacuum tower, not back to slop but to the vacuum heater inlet?
SHELTON (KBC Advanced Technologies, Inc.)
We have modeled slop oil recycle or overflash to the vacuum charge heater several times. It does not accomplish anything, except in a lube oil operation, because lube slop wax is very light and paraffinic. But in a deep-cut fuels operation, 75% to 80% of the slop oil is resid. Vacuum charge heater excess duty is better utilized by increasing feed rate or increasing heater outlet and flash zone temperatures to lift more gas oil product.
LEE (BP North America)
I will add that in our experience, we recycle the slop wax to the furnace inlet on several of our units, maybe 40% of them. However, we do find that if you are at a furnace outlet temperature limit, the slop wax recycling will cost you more duty; however, it will give you more lift. You do get a cutpoint gain, but you have to judge your unit limit.
CORY NOYES (Marathon Petroleum Company)
How do you typically set the wash rate typically? Is it done once and verified with lab results, or is anyone trying to automate that wash rate effectively based on the energy balance around the slop wax tray?
SHELTON (KBC Advanced Technologies, Inc.)
As mentioned, our models specify the overflash at the bottom of the bed, which includes the column energy balance and produces the wash rate at the top of the bed. We optimize the packing versus grid and bed height, which affects the depletion ratio. However, with some grid at the bottom of the bed, it is difficult to attain two theoretical stages because the packing height becomes excessive. In theory, we target two theoretical stages for good, deep-cut separation. In our grassroots designs, we specify radial thermowells at three levels in the wash bed. Unfortunately, after detailed design and cost cutting, very few of our units have wash bed radial temperature indicators at multiple levels. We prefer to monitor what happens in the wash zone, but there are only a few units with radial temperature indicators. We would like to see the wash zone operation optimized by monitoring temperature gradients, overflash, and HVGO yield and quality. But again, we target sufficient wash at the bottom of the bed to prevent coking.
SHELTON (KBC Advanced Technologies, Inc.)
Wash zones with 100% grid will last longer than wash beds with combination grid at the bottom and structured packing on top of the grid. For the same operating conditions, the grid will not produce the VGO recovery or the cutpoint that can be achieved with structured packing. Deep-cut designs for maximum VGO recovery are typically combination beds. There is no typical service life for vacuum column wash beds, and KBC is not aware of any vendors that provide guarantees. Crude type, severity of the operation and column design affect run lengths. The same cutpoint can be achieved at a lower transfer line temperature in a wet design with a bottoms stripper compared to a dry operation with no bottoms steam. Lube oil operations typically operate at low cutpoint with high quality, paraffinic feeds and will not be included in this discussion. Lube oil run lengths would be longer than seven years.
In fuels refineries, wash zones operate well for the normal four- to six-year run between scheduled turnarounds. For heavy crudes with high asphaltenes in deep-cut operation some sections of packing typically require replacement during turnarounds. If the question has been posed by a refinery operating under these severe conditions, a seven-year service life is better than average, particularly when processing heavy crudes at high flash zone temperatures associated with deep-cut operations.
Wash beds with structured packing over grid have wash oil depletion ratios of 5:1 to 6:1, depending on bed heights and operating conditions. Approximately 80% to 90% of the hot wash oil flow to the top of the bed will be vaporized in the bed. Targets for minimum wash oil penetration through the bed or overflash (slop oil) at the bottom of the bed are often quoted as 0.10 gpm/ft2 to 0.20 gpm/ft2 . It is important to specify if the flux rate is calculated at flowing or standard conditions. The 0.10 gpm/ft2 overflash flux rate is usually at 60°F, which is equivalent to 0.15 gpm/ft2 at operating temperature. KBC designs for 0.20 gpm/ft2 at 60°F standard conditions. Petro-SIM™ oil characterization produces the corresponding wash oil rate at the top of the bed without adjustment or design factors.
Accurate flash zone conditions are difficult to obtain for operating units. For prediction cases and vacuum unit design, KBC employs integrated VIS-SIM models of the fired heaters, transfer line and vacuum column to calculate transfer line outlet and flash zone conditions. For a specified heater outlet temperature, Petro-SIM™ accurately predicts the depletion ratio based on theoretical stages and the amount of wash oil required at the top of the bed to produce the target overflash or slop oil at the bottom and minimize coking. CFD studies show that phase separation does not occur at transfer line outlet velocities of 200 fps to 300 fps in properly designed piping configurations. Predicted flash zone temperature and pressure are a function of accurate model results for heater tube-by-tube and transfer line pressure drop profiles.
Wash Oil Coverage and Distribution: KBC specifies spray distributors designed for 200% to 300% coverage to ensure full wetting of the wash bed. Some vendors concur, and others specify trough distributors due to concerns about potential entrainment of atomized wash oil. In our experience, entrainment is more dependent on vapor velocity, the design of the flash zone inlet distributor, and the wash zone spray distributors (spray angle, pattern, elevation above the bed, nozzle type, and pressure drop). Trough distributors in the wash zone can plug due to solids and coke. Uneven level of gravity flow distributors in large diameter vessels can result in inadequate coverage of the wash bed. KBC designs spray distributors so that wash bed coverage is maintained if one of the double or triple coverage spray nozzles fails in any single spray pattern. Proper wash oil system design, metallurgy, filtration and installation can eliminate spray nozzle plugging. Although usually omitted during final design, KBC process designs specify radial temperature indicators at multiple levels in the wash bed to provide indication of flow distribution through the bed and identify problem areas.
Recommended Maximum Slop Wax Draw Temperature: The slop oil liquid temperature should be approximately 15°F cooler than the Flash Zone temperature to confirm that the lighter wash oil (HVGO) has penetrated the Wash Bed and mixed with the overflash (slop oil).
A limit should not be set on this temperature; it should be cooler than the flash zone.
BASHAM (Marathon Petroleum Corporation)
This depends on severity of the operation. As temperatures increase above 770°F, then life expectancy drops. Also, as C-factor increases, life expectancy drops. As overflash drops below 0.2 gpm/ft2 , then life expectancy drops. We have beds that have been in service for over 10 years in mild service. We have had beds in severe service that last only a couple of years. In regard to the maximum slop wax draw temperature, a primary consideration is slop wax pump reliability. To maintain pump seal integrity we recommend a maximum temperature of 700°F, which is achieved with vacuum tower bottoms quench.
LEE (BP Products North America)
We have had a grid wash bed last 20 years in Alaskan North slope service. When it was finally removed due to suspected performance issue, it was found to be in pristine condition and the bed could have gone at least another five years. We do not think a temperature limit per se is necessary, but more of maintaining an appropriate minimum bed wetting rate for the conditions and feedstock.
DENNIS HAYNES (Nalco Energy Services)
The use of KOH (potassium hydroxide) is limited compared to the more abundant use of NaOH, so there is comparatively fewer field data for KOH. It should react similar to NaOH regarding the ability to reduce chlorides for atmospheric overheads. One reason that some refiners are evaluating it is in order to reduce sodium content going to downstream conversion units, such as coker. The use will reduce sodium going downstream where there is concern in keeping this specification in greater control; however, potassium is also known to have properties (such as hydrocarbon dehydrogenation) that may increase coking. Potassium is assumed to have less detrimental properties than sodium but is not a total solution to downstream concerns.
ANDREW SLOLEY (CH2M HILL) Wash bed coking tends to be a catastrophic problem. Either the unit cokes or it does not. Many plants have achieved 20-year, or longer, service life without having a coked bed. Bed coking is caused by excessive liquid residence time at high temperatures. Keeping liquid flowing over the bed keeps the residence time down. The accepted minimum liquid rate on the bed is 0.15 gpm per square foot at the lowest liquid loading point. This will be at the bottom of the bed. Plant data has shown this liquid rate is the same for both grid and reasonably open structured packing. This value assumes good distribution of both the liquid and the vapor to the wash bed.
In operation, the plant meters or controls the liquid to the top of the bed. Liquid dryout ratios across the bed may vary from 5:1 to as high as 15:1. If the bed dryout ratio is higher than expected, the bottom of the bed will coke. This dryout ratio depends upon the pressure drop across the bed, fractionation efficiency of the bed, transfer line performance, crude composition, and cutpoint of the unit.
Very little evidence of coking will be seen for the first 10 to 14 months. After that point most units will see a noticeable pressure drop rise across the bed. However, once formed, further coking is inevitable.
A few plants chose to deliberately run with excessively low liquid rates. The objective of these plants is to tradeoff a long period with higher liquid yields (due to lower wash oil rates) versus a short period of time with lower yields due to pressure drop. The economics of this operation seem doubtful for a typical facility. The temperature difference between the slop wax collector liquid and the flash zone is more important than the absolute temperature of the flash zone. The more liquid that leaves the wash bed, the lower the slop wax temperature compared to the flash zone temperature.
The temperature difference sufficient to prevent coking will vary with the unit. The temperature difference resulting from sufficient liquid will go up if the vapor and liquid distribution to the wash bed gets worse. To maintain a minimum liquid rate at all points of the bottom of the bed, more total liquid is required.
Careful design and installation of the liquid distributor and slop wax collector tray is amply justified. So are effective control systems to monitor bed performance.