Question 26: What is your Best Practice for packing (material and shape) in isomerization unit off gas caustic scrubbers?
DUNHAM (UOP)
UOP recommends carbon Raschig rings. The key here is to specify to the vendor that this be used for caustic service so that the rings will be formed with the proper binder. If you get the wrong binder, the rings will dissolve. We have a similar service in HF alky units where we use carbon Raschig rings; and there, you have to make sure that the binder is resistant to acid. So, you cannot switch the two. I have seen the carbon Raschig rings ooze out of the tower when a customer bought the wrong binder.
Another material you can use is chemical-grade polypropylene. This is plastic, so it does have temperature limits. If you use this kind of material, you have to be careful not to steamout the tower because this packing is good for the operating temperature of the unit. But if you steam it out, it will melt, and all come out in one piece.
LAMBIE (KBC Advanced Technologies, Inc.)
My experience has only been with the carbon Raschig rings. I have not seen any issues with them in any other sites. My only other comment to add to Daryl’s is that the use of ceramic rings is not recommended because they are not compatible with caustic.
DOMINIC VARRAVETO (Burns & McDonnell)
My question is related to the scrubber. Have there been any comments from the operators of these units with upsets that cause caustic to go backwards into the stabilizer and create plugging in the stabilizer overhead system?
LAMBIE (KBC Advanced Technologies, Inc.)
I have not seen or heard of this happening. Normally there is a check valve in the line to prevent any backflow from the caustic scrubber to the stabilizer.
RICK GRUBB (Chevron Products Company)
We have experienced caustic back-flowing in the overhead receiver drum, so we have installed quick-acting ball valves that activate pressure differential. When the pressure differential gets low enough, it will close the valve.
SCOTT LAMBIE (KBC Advanced Technologies, Inc.)
One-inch OD carbon Raschig rings have been the standard packing material for isomerization unit off gas caustic scrubbers. Use of this packing material has proven to be very effective in minimizing caustic breakthrough and water carryover. Operating the unit per the design circulation flow rates of caustic and water insures proper wetting of the packing material for maximum effectiveness.
The use of ceramic packing material should be avoided as caustic will attack and degrade the material.
Question 74: In your experience, what are the effects of different Ni passivation technologies on the performance of CO promoters and stack emissions?
ZACH BEZON [United Refining Company (URC)]
Nickel passivation can be accomplished a few ways: antimony, bismuth, or sulfur. Adding antimony or bismuth in a solution, such as LCO, will immediately begin to passivate the negative effects of nickel poisoning. Antimony is considered a fugitive emission and may be hazardous; bismuth may be the safer option. Antimony decreases the effect of CO promoter; additionally, it can lead to elevated NOx emissions. Refinery fuel gas containing H2S can be used as lift gas and act as a temporary nickel passivator in the reactor, helping to reduce hydrogenation reactions. However, this effect is only temporary and will lead to elevated levels of nickel if nothing further is done.
CHRIS CLAESEN (Nalco Champion)
Pt promoters are sometimes used to oxidize CO to CO2in the regenerator. Antimony can passivate platinum promoters, which can result in afterburn and the need to use more platinum promoter. The Pt promoter oxides nitrogen compounds, as well as CO; so, the more Pt promoter that is used, the higher the NOx. Therefore, Sb (antimony) can theoretically result in higher NOx. Units using a Pt-based combustion promoter can consider a switch to one of the non-Pt combustion promoters if they want to eliminate the effect of increased NOx generation.
DENNIS HAYES (Nalco Champion)
Antimony-based additives are known to be effective at nickel passivation, but they have also been reported to reduce the effectiveness of CO promoter additives, resulting in increased usage of the additives.
ALEXIS SHACKLEFORD and BILGE YILMAZ (BASF Corporation)
Using antimony for Ni passivation will make CO promoters less effective. For example, one unit running 1000 ppm e-cat (equilibrium catalyst) Ni started using antimony, targeting a 0.35 Sb/Ni ratio. They had to increase their CO promoter additions from 1.1 ppm platinum (Pt) in inventory to 1.6 ppm Pt, or 50%. Running economics around unit profitability, the refinery found that using antimony improved profitability by 0.15 $/bbl (cost per barrel) due to lower hydrogenand coke. Even though this unit had relatively low Ni at 1000 ppm and had to use more CO promoter, the benefit of antimony at 0.15 $/bbl was significant, and they continued to use antimony.
For units that have operational challenges using antimony, Ni passivation based on catalyst technology is an attractive route. For example, the use of boron-based technology (BBT),developed by BASFand used in our catalyst BOROCAT™, has been demonstrated successfully in commercial operations to passivate Ni without impacting stack emission or CO promoter efficacy.
Question 66: Some refiners are considering substituting potassium hydroxide for sodium hydroxide as a desalted crude treatment to lower overhead chlorides. What is the impact of this change on coker operation and other downstream units? What are the advantages and disadvantages?
DION (GE Water & Process Technologies)
They are both alkali metals. Potassium hydroxide should, in theory, act like sodium hydroxide. The effect should be similar with regard to reducing the overhead chlorides in the desalted crude or, similarly, in metal-catalyzed fouling. Potassium hydroxide does have the potential to be used as a tracer; for instance, it can be injected in the desalted crude. The bottoms and heavy gas oil fractions can be analyzed for potassium to determine the disposition of adding an alkali metal to the desalted crude. Potassium hydroxide is typically more expensive than sodium.

DION (GE Water & Process Technologies)
Potassium is an alkali metal just like sodium. Its ability to reduce the overhead chlorides is expected to be comparable to that of sodium. Just like sodium, it will contribute to metal catalyzed polymerization in downstream units. There is no known advantage to using potassium other than the fact that it can be used similar to a tracer to quantify the impact of metal hydroxide addition to downstream units. Potassium hydroxide is typically more expensive than sodium hydroxide. While there are no common potassium specifications for finished fuels, its potential negative impact in the fuel should be similar to that of sodium.
BASHAM (Marathon Petroleum Corporation) Potassium hydroxide is more expensive than sodium hydroxide. It is expected that the potassium ion will catalyze the cracking reaction and promote coking in the same manner as the sodium ion. Therefore, we do not see an advantage in using potassium hydroxide.
LEE (BP Products North America)
Sodium chloride is a very stable chloride and has a very high temperature for hydrolysis. We are not sure that there is any benefit from using potassium chloride as a base to control the overhead chlorides. We think that sodium will catalyze coke formation in the vacuum and coker heaters at concentrations of over 15 ppm. We think that potassium will do the same thing. Sodium is a poison for the downstream hydroprocessing units and so is potassium. We are not sure there is any benefit here.
ANDREW SLOLEY (CH2M HILL)
Potassium hydroxide has a lower potential to induce stress corrosion cracking than sodium hydroxide. Little direct experience is available to quantify this. However, test work targeted at syngas systems showed significantly increased temperatures were required for stress corrosion cracking to occur. This may be a benefit for the crude unit between the desalter and the crude tower.
Question 4: How reliable are the dry gas seals on hydroprocessing recycle gas compressors? What are the system components put in place to enhance the reliability?
Shankar Vaidyanathan (Flour)
Dry gas seals have been used for compressors for many years. The feedback was mixed in its infancy, and there were teething problems. External factors such as the contamination of the sealing gas, insufficient sealing gas pressure and process gas leak onto the seal ring surfaces have been the main reasons for seal degradation. Wetness, particulates, and heavier hydrocarbons cause seal reliability issues. The quality of seal gas and buffer gas, dryness level, pretreatment, liquid separation and filtering are the keys to reliable operation. Experience has shown that seals can last a long time in a clean environment and many existing plants have switched over to gas seals. Tandem self-acting dry gas seal with internal labyrinths has been selected in many recent projects.
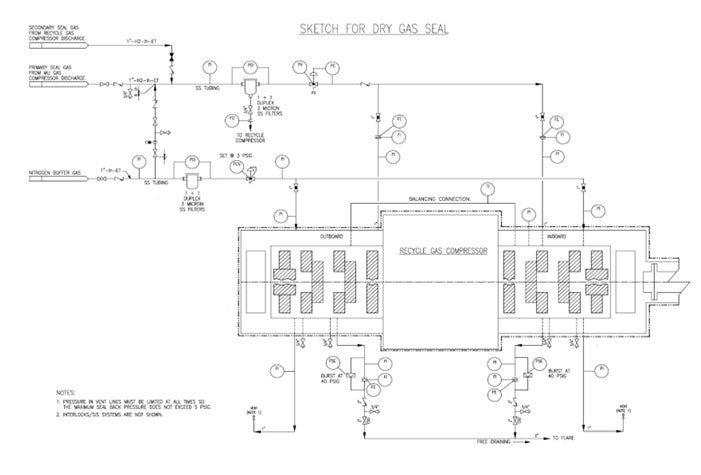
Please see the attached sketch for external components of the dry gas seal system.
1. Depending on pressure balance, the sealing gas may be pure makeup hydrogen (if the makeup compressor is located within the same plot) or recycle compressor discharge gas. Chlorides in makeup hydrogen and amine aerosols in recycle hydrogen are potential contaminants.
2. Nitrogen is used as the buffer gas.
3. Filters for seal gas and buffer gas should be duplex, one operating and one standby, 3-micron stainless steel coalescing filters equipped with high differential pressure alarm. Avoid three-way valves for filter switching.
4. Consider additional pre-filter for sealing gas if experience has shown that coalescing filters are inadequate. Consider additional demister filter for buffer gas if necessary.
5. Minimum 1” size stainless steel piping is preferred. Additional notes for piping layout include minimizing runs, avoiding pockets, heat tracing and winterizing as necessary.
6. The seal gas injection is on automatic pressure control.
7. Primary vent is routed to flare; secondary vent is routed to safe atmospheric location.
8. Primary seal vent flow is monitored with high flow and high-pressure alarms.
9. Pressure in the secondary vent line must be limited such that the maximum seal backpressure should not exceed 5 psig.
10. Consider differential pressure alarm between the buffer gas and secondary vent.
Minh Dimas (CITGO)
Dry gas seals are more reliable than oil seals provided the seal gas conditioning system is properly designed, and the seal gas is very clean and dry. That said, the filtration and liquid removal system must be very reliable and have spare equipment to maintain its reliability. Tandem dry gas seals require a source of very clean, dry seal gas at startup.





