Question 40: As it relates to overall catalyst cycle life management, please address the following issues: What are typical cascading practices for catalyst reuse after regeneration and eventual disposal that you employ? What quality control, catalyst properties and performance specifications, and/or warranties do you have in place for regenerated catalysts? What are some of the key decision criteria you use in determining whether to send a catalyst for metals reclamation, r
JAMES “TIM” CAMPBELL(Eurecat U.S., Inc.)
First, a response to the question: What are typical cascading practices that you employ for catalyst reuse after regeneration and eventual disposal? As the leading catalyst regenerator, Eurecat sees NiMo and CoMo hydrotreated catalysts (regenerated and regenerated plus rejuvenation) in ULSD, jet, kerosene, naphtha, and gas oil hydrotreating units. There is growing use of regenerated hydrocracking catalyst. These regenerated catalysts, or those regenerated and rejuvenated, may be used as an entire load or a partial load, depending on the specific application. Catalyst reuse management can provide substantial savings to the refining organization.

The second question concerned quality control, catalyst properties and performance specifications for regenerated catalysts. Eurecat has specific catalyst regeneration specifications for:
*Contamination metals (such as Si, As, Fe, Ni, V, Ca, Na),Physical (length/diameter, length distribution, and crush strength),
*Pressure Drop (measurement regenerated catalyst pressure drop versus fresh catalyst), and
*Activity (measurement of HDS activity versus fresh catalyst).

LIFENG ZHENG (Criterion Catalysts & Technologies)
Depending on the condition of the catalyst post regeneration, the catalyst can be cascaded to a less severe service [USLD to kero (kerosene), kero to naphtha, etc.] where the catalyst performance is less sensitive to activity. Regen can also be used on non-activity-constrained higher performance units or in units slated for a short cycle due to turnaround planning in, for example, the top bed of ULSD unit. In certain instances, it may be necessary to install some fresh catalyst to make up for a potential loss of activity and volume of the regenerated catalyst.
After a conventional regeneration and depending on the type of catalyst (Type I or II), the catalyst will typically regain anywhere from 70 to 95% of its fresh catalyst activity. Here your catalyst vendor can give guidance on the expected recovery for the particular catalyst. There will be physical catalyst loss due to breakage and attrition of pellets during unloading and regeneration that will need to be taken into account. Part of a catalyst bed that is unloaded may not be suitable for regen depending on the feed poisons and physical condition of the spent catalyst. Contact the regen vendor for additional details regarding warranties and catalyst properties on regenerated catalyst.
Some refiners actively manage a pool of their own regenerated catalyst because they can keep track of the condition of the catalyst based on the service and feed contaminants to allow cascading. Where it makes sense, Criterion works with the refiner to incorporate the regenerated catalyst into the planned loading for the next cycle. In a recent example, we successfully helped a customer transition catalyst from the bottom bed of a ULSD unit which was prematurely shut down into the top bed of a medium severity FCCPT unit in order to assist the customer with maximizing catalyst utilization.
The business case for selecting metals reclamation, regeneration, or disposal is ultimately based on economics. Regeneration is chosen when a use for the regen catalyst is identified within the refinery, if it is needed in the shared regen pool, or if there is a known market for this particular catalyst load and a third party is willing to purchase it. Service history is critical as catalysts with suspected high poison levels are not suitable for reuse. The catalyst cannot be vacuum-dumped, caustic-washed, or otherwise mishandled if it is going to be regenerated.
If the catalyst is not going to be regenerated, then it must either be reclaimed or disposed. Pricing and yield of precious metal could impact returns on metal reclamation. Reclamation companies usually charge a service fee for processing spent catalyst and give a credit for a portion of the metal reclaimed. Depending on the metals market, the metals credit can cover the processing fee; but with current depressed metals pricing, the credits usually do not offset the costs.
Disposal of spent catalyst is rarely done due to cost and potential environmental impacts. The transportation and disposal of spentcatalysts are governed by DOT (Department of Transportation) and RCRA (Resource Conservation and Recovery Act) regulations. The hazardous waste must be properly disposed of at an approved treatment, storage, and disposal facility.
HENRIK RASMUSSEN (Haldor Topsoe, Inc.)
For many years, refiners have cascaded used catalyst from a high severity to a lower severity service within their refinery. In order to do so, the catalyst needs to be regenerated and properly evaluated to make sure the level of poisons on the catalyst is acceptable for reuse in any service. Topsoe has, for many years, offered our proprietary ReFRESH™ technology, which is an add-on to the regeneration procedure. The ReFRESH™ process will restore the catalyst to 95+% of its original fresh activity, thus enabling the refiner to use the ReFRESH™ catalyst in the same service from which it was removed without any noticeable penalty in performance or cycle length.

In order to ensure that the spent catalyst is a good candidate for regeneration, as well as a candidate for the ReFRESH™ technology, we have generated the following guidelines. The spent catalyst should meet the following criteria:
*Surface area greater than 80% of fresh catalyst surface area,
*As(arsenic)lessthan0.1wt%,
*Pb (lead) less than 0.15 wt%,
*Na (sodium) less than 0.3 wt%,
*Si (silicon) less than 1.0 wt%,
*Fe (iron) less than 1.0 wt%,
*No other metal [Ni(nickle), V(vanadium), etc.] higher than 1.0 wt%,
*Total contaminant level less than 2.0wt %,
*Average length of particle higher than 3.0 mm(milliliters) for 1/20” TL (transferline), and
*SCS(syntheticcatalysticscavenger)higherthan2.5lbf/mm (pound-force foot to Newton millimeter).
Catalyst with contaminant levels higher than shown above should be set aside and sent for reclamation, because it is not economically justifiable to spend money on regenerating and investing in ReFRESH™ technology for this material. Every year, Topsoe applies our ReFRESH™ technology to millions of pounds of regenerated catalyst, which are used again in high severity hydro treating applications such as ultra-low sulfur diesel and FCC pretreatment. Many of our clients have used the same catalyst up to three times.
Question 36: What changes have you made to the C5/C6 Isomerization unit to comply with the new benzene regulations; what changes have you made to the refinery operation; and what have been your challenges and successes of implementing the new configuration?
Olivier Le-Coz (Axens)
More severe regulation in term of Benzene in the gasoline pool can lead to increase the Benzene content to the C5/C6 isomerization unit. This can happen in two different ways.
The refinery process operation can be modified to decrease the benzene precursors content in the heavy naphtha to the Reformer. This is achieved by increasing the light naphtha end point in the topping lights ends naphtha splitter, light naphtha being the Isom unit feed. At the same time C7+ in the Isom feed must be limited to 2 – 3 vol% as those products will undergo undesired hydrocracking reactions in the Isom reactors. With such a scheme, straight run naphtha Benzene (native Benzene) is basically treated in the Isom. This approach can typically be applied in the frame of a new project.
When the “pre-fractionation” scheme cannot be implemented or if it cannot allow reaching the overall pool Benzene specification, a “post-fractionation” option can be implemented. It consists in splitting the reformer product and recover a light Benzene rich reformate which will be treated in the Isom unit in blend with the light straight run naphtha. Depending on the Isom unit existing configuration, some modification to the hardware may be required or not. As a matter of fact, Benzene concentration at the Isom reactors inlet should better not exceed certain value to ensure proper operation and performances of the Isom catalyst (about 4 vol%).
-If the Isom unit is equipped with a recirculation, the recirculated stream acting as diluent may allow maintaining Benzene below the desirable value at the reactor inlet. The extra Benzene amount in the feed will be hydrotreated by the Isom catalyst without disturbing too much the operating conditions and without preventing suitable isomerization rate to be achieved.
-If the Benzene content at the inlet of the reactors cannot be maintained low enough (too low or no recirculation), a dedicated Benzene saturation reactor must be added.
In the case of new units implementation, those schemes have proved to work very well. In the case of revamp projects, existing equipment modifications or idle equipment reuse, a through basic design study upfront including the catalytic aspects is strongly recommended.
Brad Palmer (ConocoPhillips)
In general, refineries with C5/C6 Isomerization units or Aromatic Extraction units have increased feed rate and/or benzene content to these units. Reformer octane has gone down due to ethanol blending but, in most cases, Isom octane demand has remained strong. The primary successes include implementing these projects safely and achieving our benzene reduction requirements. Additionally, heavy reformate blend qualities have improved which has made blending premium gasoline easier and has provided additional opportunities for blend component sales.
A number of technology options were chosen by ConocoPhillips refineries to meet benzene regulations according to the existing configuration and site economics. These options include revamping Aromatic Extraction Units (AEUs) to increase feed and benzene production capacity, sending light reformate or heart cut to other AEUs, modifying C5/C6 Isom units to include benzene saturation reactors, new benzene saturation unit construction, reducing benzene production through prefractionation and use of credits.
All completed projects are working, some with very few operating problems and a few with requiring design modifications and/or operating changes. Operating, design and reliability issues continue to be worked to improve unit performance; a few specific examples are provided below.
Isomerization Unit Challenges
-When all benzene saturation reactors are complete, two will have Pt catalyst and four will have nickel catalyst. One of the reactors will have changed from Pt to Ni.
-The units that added a benzene saturation reactor in front of their Isom reactors have had challenges controlling temperatures profiles of all three reactors especially when liquid recycle is added or removed.
-Isom units have heavier feeds (increased X-Factor). One unit has and XF of 30 lv% average (35 lv% highest) and 9 lv% Benzene Average (10 lv% highest). Another unit has an XF of 25 lv% average (27 lv% highest) and 5 lv% Benzene Average (10 lv% highest).
-Determining when and how much liquid recycle is necessary for safe operation while maximizing fresh feed throughput has taken time. Vendors advertise an upper benzene level of 5 lv% to the inlet of a benzene saturation reactor. While we have gone a little higher by lowering the inlet temperature to accommodate the exotherm, this is a good rule of thumb.
-Increased unit rate can impact dryer operation by fluffing up-flow desiccant beds. Higher rates increase HCl loading to existing caustic scrubbers; less than adequate neutralization can lead to corrosion problems.
-Benzene saturation catalyst has been deactivated or poisoned by feed (organic sulfur, H2S, FeS, Chlorides) or hydrogen purity (CO and CO2) problems.
Aromatic Extraction Unit Challenges
- Changes in feed quality have required operations to find new equilibrium; one unit has reported bigger swings in aromatic content with new feed streams.
- Stripper foaming has occurred in one unit.
Ujjal Roy (Indian Oil Corporation)
In India, the benzene specification in gasoline is 1 vol.% max. In order to meet this specification, number of changes in the refinery configurations have been done. (a) Light Naphtha splitter has been introduced to produce C-5 & C-6 isomerization feed. (b) Naphtha splitter modified to reduce Benzene precursor from Reformer feed Naphtha. (c) Reformate splitter has been installed to separate Benzene from the Reformate. Over and above FCC gasoline being a component of Gasoline, for reduction of Benzene, a FCC gasoline splitter has been put to take away the Benzene rich cut called Heart Cut from Gasoline. For meeting benzene regulation in the Gasoline, Isomerisation unit has been designed with catalysts having dual functions – Isomerisation and complete saturation of Benzene. The metal sites are used for saturation of benzene and acid sites are used for isomerisation of C-5/C-6. Up to 9.8 vol.% benzene in feed, catalyst is able to saturate to nil level of benzene in isomerate.
Erik Myers (Valero)
The Valero approach has been to consolidate the benzene rich streams from various refineries and capture benzene as a product stream. This has been accomplished through use of a side draw stream from the reformate splitters and then feeding these streams through a centralized benzene extraction unit.
Question 40: Are there instances where mercaptan treatment of refinery gasoline or naphtha streams is necessary? What are the applicable treatment methods?
Praveen Gunaseelan (Vantage Point Consulting)
As mercaptans are sulfur-bearing compounds, they are one among numerous target species for sulfur removal from naphtha or gasoline streams to meet reactor feed or finished product sulfur specifications. Streams that need to be aggressively treated to low sulfur levels, such as naphtha feed to catalytic reformers, or ultra-low-sulfur gasoline product or blend stock, often require hydrotreating, which targets removal of a broad array of contaminants, including mercaptans.
However, there are a number of instances that warrant targeted removal of mercaptans species from refinery naphtha and gasoline streams (generally achieved through mercaptans extraction or sweetening). Some examples are provided below.
For light gasolines with a high proportion of mercaptans sulfur, selective extraction of mercaptans may be a competitive alternative to hydrotreating. For example, light straight run naphtha or FCC light naphtha with a high proportion of mercaptans sulfur may require only caustic extraction to be rendered acceptable as gasoline blendstock. In the case of FCC light naphtha, caustic treating for mercaptans can help avoid octane loss from olefin saturation during hydrotreating.
Light (C1-C6) mercaptans have an objectionable odor and corrosion potential and are prone to accumulate in refinery naphtha and lighter streams. In instances where naphtha is segregated, such as for use as a feedstock for downstream processing, there may be a need to reduce light mercaptans content to render the material transportable, regardless of the total sulfur content. In such instances, caustic sweetening of the naphtha may be appropriate, where the light mercaptans are oxidized to odorless disulfides.
Besides meeting sulfur specifications, gasoline streams may require meeting a mercaptans specification, such as a negative Doctor test. If the mercaptans specification is difficult to achieve through hydrotreating (for instance, due to recombinant mercaptans), mercaptans sweetening of the stream may be required.
Selective hydrotreating of FCC gasoline can result in the formation of recombinant heavy mercaptans due to the reaction of olefinic species with H2S. Depending on the sulfur level, these mercaptans may either have to be extracted (to meet the minimum sulfur specification) or sweetened to disulfide to render the gasoline acceptable as blendstock. Proprietary reagents are typically required in such instances.
For tank inventories or cargoes of gasoline or naphtha that are off-spec due to high mercaptans levels, mercaptans scavengers are typically used to treat the material to specification in a batch/semi-batch setting. Continuous treatment of liquid streams for scavengers is not typically performed because it is uneconomical compared to dedicated treatment processes.
Michael Windham (UOP)
Gasoline and naphtha streams if routed to gasoline pool should meet the following specs: Total S, mercaptan sulfur, Doctor test, CuStrip and Silver strip corrosion. If total sulfur is not required, Minalk Merox can be used to meet all of these specs. However, if total sulfur reduction is required, an extraction Merox should be used.
Of course, mild hydrotreating can also be used if reduction of sulfur is a must. However, for increased flexibility of the hydrotreating severity, a Minalk should be installed on its product.
Brad Palmer (ConocoPhillips)
Besides the obvious need to meet gasoline sulfur specifications, mercaptans tend to be malodorous and some tend to promote fuel instability by acting to aid initiation of gum formation by peroxidation. To deal with these situations, refiners can employ either mercaptan removal using strong caustic (extraction) or mercaptan oxidation that converts mercaptans in-situ to disulfides (sweetening).
Extraction is viable for the lowest molecular weight mercaptans. As the hydrocarbon chain containing the mercaptan group grows, the less water soluble the mercaptan becomes. Extraction efficiency drops off rapidly after ethyl mercaptan. Only lighter gasoline fractions will contain mainly methyl and ethyl mercaptans, (light cat or coker naphtha, C5-C7 paraffins). Heavier gasoline fractions will contain not only heavier mercaptans, but also other sulfur compounds that will neither be subject to caustic extraction nor sweetening.
Extraction can be done on a "once-through" or regenerative basis. Since extraction is equilibrium limited, once-through treating can become costly as only a small portion of the caustic value can be consumed before a significant breakthrough to the finished product occurs. Regenerative extraction processes such as UOP's Merox™ and Merichem's Thiolex™ allow the lightly loaded caustic to be reused. Distillation regeneration as well as oxidation regeneration is available, with oxidation being the most widely employed. However, distillation regeneration is not likely to be used in gasoline extraction as the extraction of heavier mercaptans will be limited by the residual methyl mercaptan content of the lean caustic from the regeneration.
Oxidative regeneration is accomplished using air and cobalt based oxidation catalyst to convert dissolved sodium mercaptide salts from the extraction into disulfide oils. The disulfide oils are nearly insoluble in the caustic and can be gravity separated from the regenerated caustic stream. Merox™ and Thiolex™ use variations of the contact, oxidation, and disulfide separation stages to accomplish extraction. Both technologies employ naphtha wash of the regenerated caustic to re-absorb trace disulfide oil that may be entrained in the lean caustic from the disulfide separation stage to prevent "re-entry" sulfur.
Sweetening is not an option for low sulfur gasolines as the mercaptan to disulfide conversion is done in-situ, that is, the sulfur content of the gasoline does not change. Sweetening can be used after extraction to aid in product stability and odor control.
Malcolm Sharpe (Merichem Company)
In the low-sulfur (< 10 wppm total S) gasoline world, there are potentially three (3) applications where wet treating can be utilized to remove mercaptans from FCC gasoline. Two of these solutions require that a FCC gasoline splitter be installed and the third removes mercaptans from selectively hydrotreated FCC gasoline.
In the case of splitter-derived FCC gasoline, the mercaptans can either, one, be extracted from the light FCC gasoline fraction using caustic-based FIBER FILM® technology (THIOLEXTM/REGEN®) or, two, be sweetened using caustic/catalyst/air-based FIBER FILM® technology (MERICATTM II) ahead of the gasoline splitter to convert the mercaptans contained in the light gasoline fraction into the heavier disulfide oil (DSO) molecule. This DSO leaves with the heavy FCC gasoline destined for the hydrotreater. The suitability of these applications is refinery-specific and is especially dependent on the light FCC gasoline cut-point and gasoline pool blending tolerances with respect to sulfur. The mercaptan extraction method (THIOLEXTM/REGEN®) can also be used to treat light straight-run naphtha subject to the same refinery-specific operating criteria.
Third, in some cases refiners may encounter recombinant mercaptan sulfur in selectively hydrotreated FCC gasoline. The presence of high levels of hydrogen sulfide and olefins at the outlet conditions of the selective reactor can lead to the formation of heavy molecular weight recombinant mercaptan compounds. Rather than increasing hydrotreater severity, at the expense of octane loss and hydrogen consumption, to battle this increase in product sulfur, it can be optimized using EXOMERTM technology which is designed to extract the recombinant mercaptans as they form. In this way operating expense and octane reduction are minimized while reaching target gasoline sulfur specifications.
Question 4: The economic benefit for propylene and amylene alkylation is improving. What considerations do you use in the feed pretreatment and alkylation unit operations before increasing these feeds?
CHRIS STEVES (Norton Engineering)
Increased processing of propylene and amylene feedstocks in alkylation (alky) units does bring challenges, but most will depend on the configuration of the existing unit and whether any of these feedstocks have been processed before.
Modification of a butylene-only alkylation unit to handle larger volumes of propylene may involve significant capital modifications to add or expand the capacity of C3 handling equipment. Examples include the depropanizer, C3 defluorinations (in HFalky units), and refrigeration equipment (for sulfuric acid alky units). With sulfuric acid alky plants, consideration will also be required for treating the reactor hydrocarbon stream before fractionation. Caustic treating systems may require the caustic circulation rate to increase by as much as twice the butylene-only rate to treat and remove esters from the reactor effluent of a propylene alky unit. In addition, the temperature required to break down these esters in the caustic treater will need to increase, potentially as much as 40°F above current operating temperatures, due to the higher stability of esters in the reactor effluent of a propylene alky unit.
In sulfuric acid alkylation units, separate reactors for propylene-rich and butylene-rich streams can help in managing acid consumption, as the different feedstocks respond differently with regard to acid consumption at different acid strengths and operating temperatures. A strategy of processing a propylene-rich stream in the high strength reactor and the butylene-rich stream in the low acid strength contactor can help to minimize overall unit acid consumption.
In addition to alkylation unit modifications for propylene alkylation, the alky feed treating will need to be reviewed to ensure that the sulfur is adequately handled and that C2is properly stripped from the alky feed stream. For addition of propylene feed, removal of H2S (hydrogen sulfide) with amine and/or expansion of the caustic pre-wash equipment should be considered so as to not negatively impact the operation of the mercaptan removal system with the production of non-regenerable sodium sulfide.
Addition of amylene to alky feed may also typically require modifications to the alky unit equipment. The extent of the modifications will depend on the desired level of amylene. Some considerations include the following:
In sulfuric acid alkylation units, amylene alkylation can be safely practiced at lower acid strengths than with propylene or butylene alkylation. With a separate reactor for amylene processing, the overall acid consumption on the unit can be minimized by allowing the final spending strength to fall lower than what would be practiced with butylene alkylation.
In sulfuric acid units, amylene alkylation is more sensitive to temperature than butylene alkylation; but with limited propane in a separate amylene reactor, the desired lower temperature may be difficult to achieve. Modifications to the refrigeration system may be required to optimize the individual reactor sections with regard to operating temperature.
In both sulfuric acid and HF alkylation, introduction of amylene feeds will increase production of isopentane through hydrogen transfer reactions (although at higher rates in HF alkylation). Removal of isopentane from alkylate may require fractionation changes in the alky unit. The isopentane production can be minimized through recycling of isopentane from the fractionation section back into the reaction zone, but this process would require additional fractionation equipment.
Amylene alkylation will also require a review of the alky feed treating system. Introduction of heavier feedstocks to the mercaptan treating section may impact the overall sulfur of the alky feed (which will then impact acid consumption), as the heavier mercaptans are more difficult to extract. Introduction of heavier feedstocks to the alky feed can also bring undesirable species into the alky feed, such as cyclopentane and diolefins which consume acid at a significant rate. While cyclopentane can usually be excluded from the alky feed via upstream fractionation, treatment of diolefins may require separate reaction systems to remove them from alky unit feed.
KURT DETRICK (Honeywell UOP)
The issues in an HFAlkylation unit are different for propylene and amylenes.
For Propylene:
The types of contaminants and the concentrations of those contaminants that must be removed in the feed pretreatment section is not much different from butylene. The one difference is that there can be some ethane and ethylene that comes in with the propylene feed. Ethane tends to act as a Non condensable and requires venting from the depolarizer overhead system, which will cause increased acid losses. Ethylene does not react with iC4 in the HF alky unit but tends to make ethyl fluoride, which will cause higher organic fluoride content in the untreated propane and resulting in higher alumina consumption in the propane defluorinations.
The operational issues with propylene are primarily increased consumption of isobutane and propane rejection. The increased isobutane consumption is due to the fact that about 20% of the propylene will undergo a hydrogen transfer reaction where one molecule of propylene will react with two molecules of isobutane to produce one molecule of propane and one molecule of isooctane (C8 alkylate). This reaction actually helps improve the alkylate octane, but it causes a somewhat higher consumption of isobutane than might otherwise be expected.
The propane rejection issue is often the controlling factor in how much propylene feed can be handled in each particular unit. There is a limit to how much propane the fractionation and stripping columns can handle, and that limit is dependent on the specific unit design. One problem that can occur as the amount of propane coming though the unit increases is that the concentration of propane in the main fractionator or isostripper overhead vapor increases, causing a decrease in the condensation temperature, and this temperature reduction can “pinch out” the overhead condenser, thus limiting the available cooling duty of this exchanger.
For Amylenes:
The types of contaminants present in the amylenes are a little different from the propylene and butylene feed. Also, the concentration of contaminants such as sulfur and diolefins is higher. These changes can require adjustment of the operation–or even the design –of the feed pretreatment units. For example, the heavier mercaptans that co-boil with amylenes have a lower solubility in caustic, and they tend to be present in higher concentrations; therefore, a higher caustic circulation rate may be required for the mercaptan extraction unit in the feed pretreatment section.
Amylenes can also undergo a hydrogen transfer reaction in which one molecule of amylene will react with two molecules of isobutane to produce one molecule of isopentane and one molecule of isooctane (C8 alkylate). As with the propylene hydrogen transfer reaction, the amylene hydrogen transfer reaction actually helps improve the alkylate octane; however, it causes a somewhat higher consumption of isobutane. The amount of amylene that undergoes this hydrogen transfer reaction depends on several factors and can be anywhere between 30% and 60%.
The isopentane that results from feeding amylenes (both in the amylene feed itself and that which is produced by the hydrogen transfer reaction) can cause the alkylate to have a somewhat higher Reid Vapor Pressure (RVP). It may be necessary to draw some of the isopentane out with the n-butane product if a relatively low RVP alkylate product is desired.
For Both Propylene and Amylenes:
The octane number –both RON and MON (motor octane number)–of the C7 and C9 alkylate that is produced is about 5 to 10 numbers lower than the RON and MON of C8 alkylate. So, higher concentrations of propylene or amylene in the feed will decrease the alkylate octane if all other variables are held constant. Of course, if the addition of propylene or amylene to the feed results in more total olefin in the feed to the unit, the isobutane-to-olefin ratio may decrease, which will cause lower alkylate octane and higher ASO production.
Question 86: What test method (e.g., ASTM D86, D1160, or D2887) do you currently use to determine the distillation of FCC gasolines, cycle oils, and fractionator bottoms?
AVERY (Albemarle Corporation)
I put distillation methods into two different categories. One is simple distillation or acts of distillation, which is either a D86 or D1160. D86 is at atmospheric conditions; the D1160 would be at a vacuum. More people are switching to simulated distillations or using a GC (gas chromatography) to do that. The most common number is a D2887. There are other numbers, depending on if there is oxygen in the stream, if you want light ends, or if you are really focusing on heavy ends.
The GC methods are becoming more common because they require a smaller sample size and because an operator can do far more methods that way. As an example, in our laboratories, we used the D1160s but have now switched to D2887 for most types of streams. We also use high temperature simulated distillation for some heavy resid types. We could run about 12 in a day if we did a D1160. Since we switched to simulated distillation with GC methods, one operator can do a hundred samples in a day by himself. So, we saved quite a bit a time when we switched to using GC methods.
I took a survey of over 60 FCC units worldwide. The slide shows a list of the naphtha streams, LCO streams, and slurry in feed. As you can see, D86 is more commonly used for the light boiling point ranges. This survey also ranged from 2000 to 2012. If I did a poll through that period of time, the ones close to 2000 used more of the simple distillations, like a D86, D1160; most common are all of the GC methods. More people are using what I listed as HTSD, which is high temperature simulated distillation. HTSD is a GC method that helps you measure the really heavy components that boil up to 1350°F.

I spoke with our R&D (Research & Development) guys and asked them to create a graph showing some examples of a D86, D1160, and simulated distillation. The yellow stream represents D1160 as the standard done in a vacuum. If you run a D86, you are going to see more thermal decomposition occurring above 650ºF. I forgot to mention that a D86 is generally not used for heavier boiling points; because somewhere around 600°F to 650°F, you will see thermal decomposition. You can see the D86 represented by the pink line. Once it gets into a higher temperature, about 700ºF, it will have a different slope due. When you start going to GC methods like the D2887, you will see larger tails. So, if you look at D1160 and at the simulated D2887, you will see a larger tail here in green and also a larger tail on the backend.

KEVIN PROOPS (Solomon Associates)
To the panel members in operating companies, as you have changed your distillation methods over time, have you gone back and updated all of your unit monitoring calculations to now calculate standard conversion at a 430 cutpoint using your new methods, or did you ignore the change in distillation method?
AVERY (Albemarle Corporation)
Data from refineries is often generated by using kinetic modeling. Within that process of collecting data, they will have inputs for the method. Those systems will automatically take care of the corrections.
BROOKS (BP Refining)
Our sites use models similar to those Cliff just mentioned.
SCHOEPE (Phillips 66)
I second that for Phillips 66.
AVERY (Albemarle Corporation)
Basically, distillation methods are in two categories: simple batch distillations (D86/D1160), and simulated distillations by gas chromatography. Initially refiners used ASTM D86 to determine oil boiling points (BP). The D86 simple batch distillation, or low efficiency distillation, is performed at atmospheric conditions. Decomposition or thermal cracking of the material can occur at temperatures greater than 650°F (344°C). Due to this high temperature decomposition, the ASTM D1160 method was developed. The D1160 is performed at reduced pressures [typically around 10 mm Hg (millimeters of mercury)]. At this pressure, oil fractions up to 1,000°F (538°C) can be accurately analyzed.
Simulated distillations (SimDists) are performed by GC. The most popular method is ASTM D2887. D2887 determines the boiling point distribution by injecting the oil sample into a GC that separates the hydrocarbons in a boiling point order. The retention time in the GC is related to the BP through a calibration curve. Recently, newer GC methods have been used. High temperature simulated distillation (HTSD) can accurately measure BPs greater than 1,000°F (538°C).
Simulated distillations save a significant amount of time, utilize less manpower, and require a smaller sample size: all advantages over simple distillations. At Albemarle, we run a standard D2887 for lighter BP fractions. For heavier oils such as FCC feed, we utilize a customized HTSD to assure we accurately measure the heaviest BP compounds. Our D1160 capacity was approximately twelve samples/day using one liter of oil and at least one technician. Our D2887 system has a capacity of 100 samples per day using less than one milliliter per sample and one technician.
Data from over sixty FCCUs were reviewed. The data runs from 2000 to 2012 and comes from every WW region. The general observation is that the method analyzed on feed is also performed for the slurry. For naphtha and LCO, many refiners are staying with the D86 method. More recent data and data from larger and/or major oil companies tend to utilize GC methods. Second-generation GC methods (HTSD) are showing up more often.
SCHOEPE (Phillips 66)
All Phillips 66 sites except one use simulated distillation methods for gasoline LCO and slurry distillation. D7096 is used for gasoline, D2887 is used for LCO, and high temperature D7169 or D6352 is used for slurry. One Phillips 66 site still uses D86 for gasoline and LCO.
Question 9: What has been your experience with antimony and phosphorous poisoning on hydrotreating catalyst performance? What is the maximum level?
Kaspar Vogt (Albemarle) Antimony (Sb)
The effects of antimony in oil on hydrotreating catalyst have not been directly studied, but we can infer the likely impacts of antimony from a variety of information sources and past experiences.
As background, contaminant metals such as nickel can deposit on the FCC catalyst. This will result in increased dry gas (H2 in particular) and delta coke. Depending on the unit constraints this can lead to lower FCC conversion and lower feed rate. Many refiners use antimony in the FCC riser to passivate the detrimental effects of nickel. Antimony will cover the nickel enriched catalyst surface. Side effects are that the Sb will also cover the CO and NOx promoter metals and make these additives less effective.
Excess antimony mainly accumulates in the FCC slurry. However, antimony can be present in the heavier FCC products which are hydrotreated downstream. If the antimony enriched FCC catalyst fines are entrained into the hydrotreater, they can deposit in the catalyst interstices. This will impact bed pressure drop but not catalyst activity. The bed pressure drop build up can be managed by a guard bed catalyst system of sized and shaped catalysts to increase the void fraction and create more particulates capacity.
By analogy with the FCC experience, we would expect antimony in oil to preferentially coat nickel and cobalt promoter metals on the NiMo and CoMo catalysts. Ultimately, this would completely poison the catalyst. During the buildup of coating/poisoning, the activity will likely see a shift towards direct desulfurization (DDS) vs. indirect/aromatic saturation, thus the hydrogenation-to-hydrogenolysis ratio will change. A given concentration of Sb on catalyst would be expected to have a more severe effect on the catalyst performance in high severity HDS/HDN operations like ULSD and hydrocracker pretreat (HC-PT) service than in lower severity hydroprocessing applications such as NHT and LSD.
We seldom, if ever, detect antimony in the interior of spent hydrotreating catalysts where it would be expected to impact activity.
Furthermore, given its position in the periodic table, we would expect that Sb attacks the catalyst's active (NiMo and CoMo) sites, and that it would be a relatively severe poison, similar to arsenic (As), sodium (Na) and lead (Pb). Therefore, we would expect ≤1.0 wt% Sb would reduce HDN/HDS relative volumetric activity (RVA) by approximately 50% in non-severe applications, and that even lower Sb concentrations could severely reduce catalyst activity for high severity operations like ULSD and HC-PT.
Phosphorous (P)
Phosphorous (P) can come into the hydrotreater feed from:
- crudes
- drilling fluids
- phosphated ZSM
- phosphorous-based corrosion inhibitors and flow improvers
- phosphorous from solid phosphoric acid catalyst
-biofeeds
In catalyst manufacturing, phosphorous added on hydrotreating catalyst acts as a promoter and provides additional acidity to enhance HDN, hydrogenation and cracking reactions. Phosphorous also improves metals dispersion on the catalyst surface.
In one instance, we saw that 3 wt% of phosphorus on the catalyst terminated all the exotherm in bed, although other poisons where also present. Organic phosphorous can penetrate into catalyst pores. In general, our understanding is that the poisoning was similar to sodium where ~1.0 wt% concentration halves the catalyst activity.
In a separate instance, we found SiP coming from a solid phosphoric acid catalyst, used in certain FCC gasoline desulfurization processes with some iron deposited at the external surface of the catalyst extrudate. Some phosphorous and silicon also penetrated the catalyst pores. However after the first 0.1 mm, no contaminant phosphorous and silicon were found on a main bed CoMo catalyst. In this case, a layer of P-Si-Fe had deposited at the pore mouth and restricted the diffusion into the catalyst.
Photos of the outer surface including chemical composition are shown below. They show that Alumina, Molybdenum and Cobalt are homogeneously distributed within the catalyst particle, while phosphorous, silica and iron are located at the outer surface of the particle.

We observed that the Si & P from this process behaves totally differently from Si from anti-foaming agents. There are Si-P particles which cannot penetrate the internal pores of the catalyst and are deposited on the catalyst outer surface. The accumulation of these particles cannot be prevented. Therefore, sooner or later, bridges from particle to particle are formed, thus causing pressure drop buildup.
The bottom line is that the quantitative effects of phosphorous on hydroprocessing catalyst performance and the maximum allowable levels are highly dependent on the source and form of the phosphorous compound. It is also dependent on catalyst properties and the process application.
Martin Gonzalez (BP)
Phosphorus can sometimes be found in crude as alkyl phosphates added to passivate metals or protect against naphthenic acid corrosion. Phosphorus esters in crude may originate from waste oils, or from additives injected into wells to improve recovery. Some of the phosphorus may be in a form that volatilizes into distillate fractions bound for hydrotreaters. We have encountered some Canadian crudes containing phosphorus originating from fracturing fluids used in production. Phosphorus content in light sweet crudes seems to be declining, but it may be becoming more prominent in heavy crudes. There have been reports in the industry of ULSD units suffering catalyst deactivation as result of phosphorus from these crudes. From our experience, at 1 wt% on catalyst, it is reasonable to expect a 15-30% activity loss.
Charles Olsen (ART)
Phosphorous (P) contamination in oil has been traced to frac fluids that are often used in crudes from the Western Canadian Sedimentary Basin. The source is diphosphate esters which are soluble in the crude oil. Refineries that run large percentages of light Western Canadian crude have reported crude column and crude furnace fouling for many years. Improvements made to crude columns to minimize fouling have transitioned the depositing of phosphorous to the downstream hydrotreaters.
Other sources of phosphorous include gasoline slop tanks, imported feeds and lube oil wastes. If phosphorous does manage to make its way into the hydrotreater it will poison the active sites of the catalyst causing a loss in activity. A level of 1 wt% of phosphorous on the catalyst results in roughly 10°F loss in activity. ART recommends that a feed content of < 0.5 wppm be maintained whenever possible as well as the use of feed filters to assist in trapping of phosphorous sediment.
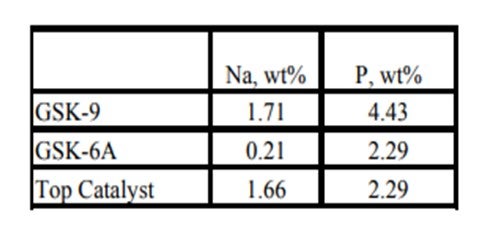
Historically, phosphorous contamination has not been very common, but with the increasing use of opportunity crudes it is being observed more frequently. A recent example is summarized in the table below shows the results of some spent catalyst analysis from a diesel unit. This unit experienced extremely rapid catalyst deactivation shortly after start up. It was so severe that within several months the unit required an unplanned turnaround and fresh catalyst was installed. The spent catalyst analysis indicates the catalysts were exposed to high levels of several poisons including sodium and phosphorous. The contaminants penetrated well into the catalyst bed. The level of contaminants indicates the catalyst in the top half of the bed had lost over 60°F of activity.
Question 26: When you test for free HF and organic fluorides in alkylation unit products (alkylate, butane, propane), what are your typical observed levels? After HF breakthrough in our butane product, why does our treater still have plenty of KOH remaining? Is there any way to regenerate KOH during the run? Do others maintain a heel of KOH in the bottom of the alkylate storage tank to neutralize traces of HF?
Erik Myers (Valero)
These series of questions overlap quite a bit. The following answers address each question in the approximate order of those questions.
Our sites vary in the type of testing with most sites testing for combined (organic) fluorides in at least the propane and butane streams. Multiple stream points are typically tested dependent on what the monitoring goals are. Typical levels upstream of any treating are:
• Propane - 200 ppm
• Butane – 600 - 1000 ppm
• Alkylate – less than 100 ppm
Combined fluorides measured upstream of treating can be used as an indication of the completeness of the alkylation reaction. There will always be some level with the typical values noted above. Higher levels indicate potential issues with the upstream operation. The above values are for typical operations. Key contributors to increases in combined fluorides are low acid strength (below 85%), low reactor temperature (less than 80 o F), decreased contact time and low I:O ratio. Any of these can lead to increases in the amounts of all levels of combined fluorides. Propyl and butyl fluoride can increase by orders of magnitude with low acid strength. Post treatment levels of combined fluorides should be well under 10 ppm, typically.
Defluorinators are typically installed on the propane and butane streams, followed by KOH treating. These systems are occasionally used on the alkylate product stream. Water and HF are the products of the defluorination reaction. HF reacts with the defluorinator alumina to make aluminum fluoride trapped as part of the defluorinator alumina. This leads to potential of trace HF in the defluorinated stream if the remaining active alumina does not convert the HF. There is typically a lead – lag arrangement on the defluorinators to allow continued treating of the product streams. The downstream KOH treater is installed to dry the defluorinated product and remove any trace HF. It is less common to have a lead – lag for the KOH treaters but two of our sites have this arrangement on at least one stream. Some of our sites have water collection pots upstream of the KOH treaters to lessen the load of those treaters. To measure the effectiveness of the defluorinators and as an aide in determining optimum change out frequency, the streams are ideally measured before, between and after the treaters (with the downstream measured after KOH treating). The sample between the lead and lag defluorinators is used for confirmation of whether an alumina changeout is required. The upstream sample, along with the product flow rates can be used a predictive tool in scheduling lead defluorinator changeouts. The spent alumina can also be sampled and analyzed by the alumina supplier and compared to these predictive results for further alumina changeout optimization as well as verification of the hydrocarbon stream fluoride testing. One site uses a typical fluoride concentration and then a throughput totalizer to determine changeout timing, then analyzing the spent alumina to confirm loading.
Aside from the trace HF noted from the defluorination reaction free HF should not exist in the alky propane if the HF Stripper has adequate reflux and never show up in the normal butane or alkylate product. The primary cause for free HF is spent alumina in the defluorinators or severe loss of tower temperature profile in the alkylation fractionation tower(s). less than 1 ppm. Only one of our sites typically checks for free HF with the values being less than 5 ppm.
The KOH treater is typically a walnut bed downstream of the defluorinators. Our sites utilize both walnut and flake KOH, with walnut being typical. Our units are split with 50% have upflow and the other half downflow. This is typically an indication of the original unit licensor design. As noted earlier, water and HF are the products of the defluorinator. If the there is an HF breakthrough to the KOH treaters, it is most likely due to a spent defluorinator, where there is no more alumina to react with the HF. Significant breakthrough is important to avoid. Large amounts of free HF can cause the KOH treater to heat up resulting in hydrocarbon vaporization and unfavorable conditions for HF removal. (In a propane KOH treater, melting of the KOH and then freezing it in the outlet piping has actually been observed). One of our sites has an emergency alarm for high butane KOH treater outlet and delta temperature with another site having an SIS diversion for high C3 KOH treater temperature. The noted upstream and downstream sampling of each defluorinator is a key to staying on top of this processing area of the alky.
If KOH is still present in the treater while HF is measured in the product it is most likely caused by poor distribution through the KOH bed, either from channeling or crusting on the top of the KOH bed, sometimes caused by low amounts of water in the feed to the KOH treater. This low water content prevents the removal of KF (formed by the reaction of the KOH and the HF) from the KOH treater. Our sites have utilized either routine steam or water injection to the KOH treaters to prevent this.
Circulating KOH (typically used in the acid relief system neutralization system) can, and typically is, regenerated in a batch mode. We are not aware of a method to regenerate the solid fixed bed units as the KOH is converted to water and drained from the system. Three of our sites have two KOH treaters (either in parallel or series), allowing monitoring and changeout to be accomplished without compromising product quality. Residual KOH / water from the KOH treater changeouts can be utilized for make up in the circulating KOH system noted above.
Only two of our sites presently utilize a caustic heal in the alkylate product tank. This has been utilized at other sites in the past. This was done either as a preventative measure or as a result of previous issues with tank bottom corrosion. It is a common recommendation from the licensor. If this method is used, the tank water draw should be monitored frequently to measure changes and prevent loss of protection. The mechanism for tank bottom corrosion is either HF breakthrough from slumping of the fractionators, an exchanger leaks that routes acid to the tank or water in the alkylate product tank that leads to hydrolysis of the combined fluorides in the alkylate to HF if the residence time in the tank is long enough. The noted monitoring of any water draws and then ensuring that there is not water is another preventative measure for this.
Brad Palmer (ConocoPhillips)
Typical organic fluoride levels in alkylation unit products, upstream of any post-treatment, have been reported as 40-60 ppm (Alkylate), 200-400 ppm (Butane), and 300-600 ppm (Propane). Inorganic fluorides are not typically measured. Defluorination and KOH treating will reduce propane and butane organic fluorides to 10 ppm or less. Inorganic fluorides will be less than 1 ppm after treatment. Thermal defluorination, occurring in the heater passes, can further reduce organic fluorides in alkylate. Maintaining the fractionator bottom temperature above 320°F will thermally defluorinate any organic fluorides in the tower bottom thereby minimizing organic fluorides in the alkylate.
Un-used KOH material at breakthrough signifies bed channeling and/or a very dry system that allows KF to coat the KOH material. Defluorinator chemistry reacts organic fluorides with alumina to form alumina fluoride and water; an intermediate reaction product is HF, which may leave the defluorinator unreacted. The KOH treater is primarily a dehydrator and secondarily an HF neutralizer. As the KOH dries the LPG stream, the water "cleans" the KOH as it makes a sludge that is drained from the vessel. Any HF breakthrough from the defluorinator will react with the KOH to form KF and H2O. If there is very little organic fluoride to react in the defluorinator, there will not be much water formed to slough the KF off the KOH pellets. Some sites have used water injection to help "clean" and utilize the KOH material under dry conditions.
There is no effective way to regenerate solid KOH in the KOH treater with the vessel on-line. Water injection might be effective to refresh KOH that has been coated with KF as previously described.
It is a common practice to use an alkali heel in the alkylate storage tank. This is not for neutralizing HF, but is to counter-act iron fluoride scale leaving the process with alkylate which can form low pH hydrates on the tank bottom. The alkali heel should be tested routinely to ensure it remains basic.
Question 46: Silicon uptake on hydrotreating catalysts is an increasing problem. (1) What operating conditions favor maximum silicon pickup by the catalyst? (2) Are there differences between silicon from coker antifoamsand other sources? (3) Does the presence of other contaminants such as nickel and vanadium affect the silicon pick-up by the catalyst? (4) What best practices are you using to monitoring silicon pick-up by the catalyst?
James Esteban and Jeff Pro (Criterion Catalysts & Technologies)
Silicon in feed streams to Hydroprocessing units can pose a threat to catalyst performance and must be properly managed. Silicon acts as a poison to the catalyst by depositing on the surface of catalyst particles blocking active sites and reducing critical HDS and HDN activity. Silicon can be found in a wide range of feed streams and is a concern for all hydrotreaters processing naphtha, distillates, and vacuum gas oils. Silicon is present in crude fractions as well as Coker feeds where Si-containing anti-foam additives are widely used. Si from crude fractions is found in higher concentrations in synthetic crudes which have been manufactured at upgrading facilities which employ the use of delayed coking processes that use Si-based anti-foams. Regardless of the source, the methods employed to remove silicon are similar. Synthetic crudes can also contain Si from sand and aluminum silicate clays. In order to properly protect active catalyst beds from Si poisoning consideration must be given to the process conditions, catalyst selection, as well as feed components. In general Si uptake is maximized by operating at temperatures above 550 F with peak uptake performance above 600 F.
For units in naphtha service the temperature regime may limit the uptake capacity of the lead catalyst beds especially when considering units that have low temperature di-olefin reactors. In distillate and heavier service, the typical operating temperature regime is high enough to support maximum Si uptake performance. Another process condition impacting the Si uptake of a catalyst system is space velocity. Units that operate at high space velocities see a lower efficiency in terms of overall Si uptake as a percentage of maximum saturation capacity due to the high space velocity stretching the distribution profile of Si in the catalyst bed.
Catalyst properties such as surface area and particle size play a key role in the Si uptake performance of the catalyst system in lighter feeds like naphtha and distillate boiling fractions. In the gas phase, as in NHT (naphtha hydrotreating) service, catalyst surface area is a critical property that determines the catalysts' ability to uptake Si. Higher surface area catalysts will have a higher Si uptake capacity for NHT service; however, they will typically have less overall HDS/HDN activity due to a limited number of active metals present on the catalyst. This is of particular concern in units that operate at high space velocities with limited catalyst volume. In these cases, dual-function catalysts that have high Si uptake capacity in addition to high HDS/HDN activity can be employed to provide the required balance of Si uptake and activity. In addition to surface area, catalyst particle size is an important factor to consider. In NHT service the rate limiting step is diffusion, which implies that smaller particle catalysts will perform better than larger particle catalysts in terms of Si uptake. The drawback to smaller catalyst particle size is a potential increase in pressure drop.
Units processing distillates benefit from the same catalyst properties but tend to be less affected by space velocity since these units are typically larger and hence have lower space velocities than naphtha units. Distillate units typically processing SR and light coker gas oils also do not have as high a feed Si content as naphtha units (especially naphtha units running high percentages of Coker naphtha). Units processing heavier feeds often contain other poisons such as Ni and V and hence require additional functionality. The catalysts also need sufficient active metals to promote the HDM reactions required for these larger molecules. Ni and V in high concentrations can reduce the Si uptake of trap catalysts; however, the Ni and V uptake is typically of greater concern.
For all applications, care should be taken to apply the appropriate catalysts for the service to optimize metals uptake with activity requirements.
In terms of monitoring Si uptake and performance one must employ a complete cradle to grave approach. Initially the catalyst system must be designed to ensure that there is adequate Si uptake capacity. With limited information in the design stage, employing the use of proper efficiency factors is critical to prevent Si slip to product streams above the desired specification. In some applications Si slip can be absolutely detrimental such as a NHT upstream of a catalytic reformer which uses very costly platinum promoted catalyst that can be poisoned by Si. Alternatively in other processes some Si slip to product is acceptable such as the treating of fractions for the blending of pipeline quality synthetic crudes. It is critical that refiners work closely with catalyst suppliers to ensure that objectives are clear, and the proper approach is applied. It is a best practice to refer to proven commercial performance when designing an optimized system and is best to make use of unit specific performance when available.
During the catalyst cycle, the catalyst activity and Si uptake should be continuously monitored during periodic unit performance reviews. It is best practice to monitor feed Si content using a method such as routine composite samples – these results can be used to calculate a projected Si accumulation which can be tracked against the maximum uptake capacity. Following the completion of the catalyst cycle spent catalyst samples should be collected to provide insight on actual catalyst performance versus predictions as well as to develop a Si distribution profile and material balance across the reactor and validate the accuracy of the composite feed samples. This methodology was well documented in an article – “Estimating silicon accumulation in coker naphtha hydrotreaters.” [1]
[1] Thienan Tran, Patrick Gripka and Larry Kraus, “Estimating silicon accumulation in coker naphtha hydrotreaters”, PTQ, Catalysis 2012.
Brian Watkins and Charles Olsen (ART)
Silicon is probably the most widespread catalyst poison encountered in hydrotreater feeds. The main source of silicon is from delayed coker operations that use an anti-foam agent based on polydimethylsiloxane to suppress foaming in the coker drums. The siloxane complex breaks down in the coking process to primarily cyclic methylsiloxane trimers. These species are volatile at coker temperatures with boiling points ranging from 270-475°F (132-246°C). As a result, these compounds tend to concentrate in the overhead products, and as a general rule of thumb, 70-80% of the silicon at the coker ends up in the coker naphtha fraction. More recently, even refineries that do not have cokers are experiencing silicon poisoning of hydrotreating catalysts once thought unlikely since their feed source comes directly from the refiner’s crude unit. These refineries have begun processing synthetic or other opportunity crudes and the process of making synthetic crude often involves a coking step. In addition, it is becoming more common to use silicon additives in the drilling process, and for pipeline companies to use them for both flows enhancing performance and foaming issues. It has also been found that silicon additives are sometimes used in barge unloading.
In the hydrotreater, the silica fragments from the antifoam agent undergo a condensation reaction with the alumina surface of the catalyst forming a strong chemical bond. Once the silicon is bound to the alumina surface, it cannot be removed by regeneration or other means. It is a more moderate poison compared to contaminants like sodium or arsenic, but it nonetheless results in activity loss of the order of 5-10°F (3-6°C) for each 1.0 wt% Si deposited on the hydrotreating catalyst.
A variety of analytical techniques have been applied to silicon poisoned catalysts, and this confirms that the silicon is associated with the alumina support as opposed to the active metal sulfides of the catalyst. Furthermore, the silicon is dispersed throughout the available alumina surface as opposed to poisoning only the exterior of the catalyst pellet. As a consequence, the available alumina surface area of a catalyst has a significant impact on silicon capacity of a catalyst.
Another important aspect of silicon poisoning is that silicon picks up depends on unit operating temperature. Commercial data clearly show that the operating temperature of the application must be considered when discussing silicon pickup capacity and when designing effective guard catalyst systems. The maximum capacity of the catalyst needs to be considered as well as the capacity at the operating temperatures of the specific unit in order to accurately predict the point at which silicon will breakthrough into the next bed of catalyst or refinery unit.
Accurately measuring silicon in naphtha streams can be done but it takes a bit of work to get a representative sample of the naphtha. The silicon in the coker naphtha depends on the type and amount of antifoam chemical at the delayer coker unit. Delayed cokers have cycles ranging anywhere between 8 – 24 hours. The coker unit is continually producing a coker naphtha stream during these cycles which is typically being sent from the fractionator straight into the naphtha hydrotreater feed drum. The antifoam chemical is usually not added for the entire coker cycle. This means that the silicon in the naphtha stream will vary with the timing of the coker cycle. In order to get a representative amount of silicon in the coker naphtha stream a composite should be made of hourly samples mixed together for the time of the cycle. For example, for an eight hour cycle eight samples would be mixed and the composite sample analyzed for silicon. To measure the silicon an ICP-MS (Inductively Coupled Plasma Mass Spectrometry) instrument can be used. This instrument/method can measure very low metal concentrations.
Question 83: Can a slurry pump run at or below 1000 rpm (revolutions per minute)? If not, what is the lowest speed to minimize pump erosion?
RUSSEFF (CVR Energy, Inc.)
The short answer is “Yes”, but it would not be a good FCC answer if I did not say the words, “It depends.” The actual effective service life depends on your solids loading in your slurry system, in both typical and upset conditions. A good set of suction strainers and coke catchers in the entire level will certainly help you. I understand, from at least one of the major licensors, that there is typically a low limit of about 1200 rpm for new units. Can you run lower? Certainly, but there are also some considerations to contemplate.

This slide shows a very old pump life curve from the old Lawrence literature, which I obtained when I was in my position. You can see “1200” annotated in red. You can also see that all of the curves start sloping up and that the coded pumps definitely have a distinct advantage. The CA6NM is uncoded, but it is also represented there without slurry contamination in its process.

So, can you run lower? Yes. What is not shown up here is the type of contaminant; that is, what was the ash or the BS&W on the particular sample? It does not tell you the nature of the component. Although it is not shown on the slide, we would expect the shape of the curve to be the same. What is also not on the slide and which is part of the “it depends” part is the information about the location of your efficiency on the curve. You know, maybe that does not matter to you, but certainly what does matter are the minimum velocities through your exchangers. So those are some of the other variables to consider. Can you run lower? Certainly. Then you will see some great lifespan there, but it is a balancing act.
SINGH (Indian Oil Corporation Limited)
Slurry pumps are extremely critical for the performance of the MCB (main column bottom) circuit. While it is essential to maintain a certain minimum velocity in the slurry circuit, the speed of the slurry pump is typically governed by the desired capacity, head, and NPSH (net positive suction head). In all of our units, the slurry pumps typically operate between 1100 and 1400 rpm; and in most cases, these pumps are fully lined with 25% chromium (Cr). A word of caution while using fully lined pumps: They are very, very sensitive to thermal shocks. Our experience with these pumps has been extremely good, as far as erosion is concerned. Even under very bad conditions with very high BS&W due to occasional high catalyst carryover situations, these pumps have been able to perform well. But like I said, they are extremely sensitive to thermal shocks, so adequate precautions need to be taken. We have also had some bad experiences caused by thermal shocks to the pumps, which lead to sudden and total failure of the liner.
We have modified the warming up system of slurry pumps to enable heating at a very controlled rate (less than 50°C (82°F) per hour) by mixing hot and cold streams. This has eliminated the problem of thermal shock and substantially improved the reliability of these pumps. A maximum 50°C (82°F) per-hour rate is usually recommended for heating these pumps. One word of caution: It is not only the pump that has to be heated; the suction line or whatever is the stagnant material in the standby pump’s suction line also needs to be heated. Otherwise, whenever the standby pump is put to service, the pump will see an unexpected thermal shock.
ROBERTSON (AFPM)
Those were the responses from the panel. Any comments or questions from Kevin Proops? [Laughter]
KEVIN PROOPS (Koch Industries, Inc.)
I am not selling pumps either. [Laughter] I will put my Pete Andrews hat on here for just a second and talk about fundamentals. Pump rotation speed is important, but it is really the impeller tip speed that will cause erosion. If you slow the pump, you will need a bigger impeller for the same discharge pressure. The curve on Richard’s slide shows 286 feet of head, which is nominally a 100 psi differential. If you really want lower erosion, can you design a system that is more like 50 psi differential? You could put all the heat exchangers in parallel. I have seen that done before. You can avoid high pressure drop control valves in the circuit by diverting the pumparound return to the bottom of the tower to control heat removal, rather than throttling.
I have seen vertical steam generators that basically do not foul. These generators have the channels on the bottom with a vertical bundle. In any design, exchanger tube velocity is important and has been discussed in past sessions. Keep piping runs short. Minimize the pressure drop in the system, and you should be able to get there.
SANJIV SINGH [Indian Oil Corp Ltd. (IOCL)]
Slurry pumps are critical for the reliability of the MCB (main column bottom) system of an FCC unit. Desired velocity in the slurry exchangers also results in a high pressure drop, setting the operational requirement for the slurry pump. Impeller diameter or the speed need to be increased to match the hydraulic requirement of slurry circuit. The speed of the pump is mainly governed by operating parameters such as capacity, head, and NPSH. Adequate minimum tube velocity in the slurry circuit is absolutely essential to avoid excessive fouling in these exchangers. In the case of slurry pumps, a lower speed will mean less erosion but will require a bigger pump size. A higher head will require a higher speed. A speed limit of about 1500 rpm has been specified typically for FCCU units. Degradation of pump performance will increase the rate of exchanger fouling and limit MCB heat removal. To minimize pump erosion from catalyst and coke particles, an API fully lined pump is often required when operating with a higher pump head and operating speeds in the MCB circuit.
Apart from lowering speed, another way of reducing erosion is to specify a fully lined slurry pump with a 25% Cr iron liner. These pumps can be operated continuously for a considerably longer period compared to a normal API pump. In all of the recent projects, licensors specified fully lined slurry pumps and the same were used with satisfactory performance. Of course, speed was also limited, but physical pump availability at that speed has to be ascertained.
For most of our FCC/RFCC units in IOCL, slurry pump speeds are in the range of 1100 to 1400 rpm. Even for this operating range, fully lined pump internals are used (25% Cr). Though these pumps have demonstrated reasonable resistance to erosion, but these pumps remain extremely sensitive to thermal shocks. Recommended warming or heating up procedure for these pumps should be strictly followed. In our newer units, we have installed facility to heat up these pumps in a very controlled manner by mixing two streams of different temperatures and fine controlling the heating rate. Further reduction of pump speed below 1100 rpm is also possible considering acceptance of the bigger size of the pump model, as well as the availability of the PTR (performance track record).
Erosion is no doubt a function of speed: the higher the speed, higher the erosion, which will require more frequent overhauling.
CHRIS STEVES (Norton Engineering)
Slurry pumps are designed to operate at low speed to minimize corrosion but trade off hydraulic efficiency to do so. Pump manufacturers with long-term slurry experience can provide proven erosion-resistant metallurgy options (casing, liner, wear rings, etc.), which allow operation of slurry pumps at relatively high speeds (1200 rpm typical) yielding hydraulic efficiency and ultimately power reduction benefits versus low speed pumps.
ANDREW W. SLOLEY (CH2M HILL)
Refiners have reliably run API pumps continuously at speeds of 900 rpm and below. The lower speed is often compensated for by using a larger impeller. Slower pump speeds are applied in most cases to reduce suction-specific speed problems. However, slower speeds also reduce erosion, as well as reducing discharge pressure and pump efficiency.
