Question 85: What is the typical range that you employ for iron content on FCC equilibrium catalyst? What methods are available to determine how iron is accumulated on the catalyst surface? How does the distribution of iron on the catalyst surface impact the FCC operation, yield structure and emissions?
Jeff Lewis (BASF)
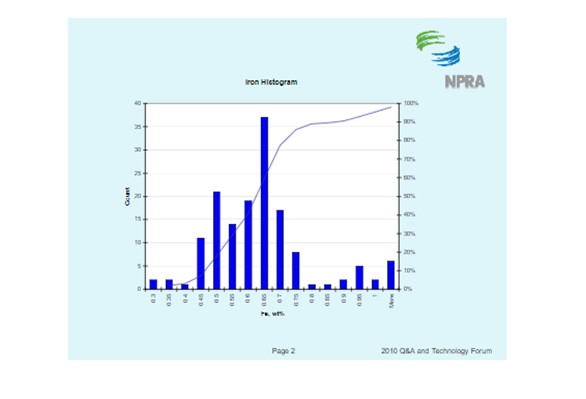
The histogram below shows the distribution of iron content for all ecat samples BASF receives. It should be noted that fresh catalyst has an iron content of about 0.55 wt%. The histogram shows that the median ecat iron concentration is approximately 0.62 wt%. This suggests the median contaminant iron level on ecat is 0.07 wt%.
There are several methods available to quantify iron contamination on catalyst. Scanning Electron Microscopy (SEM) pictures are a valuable means to qualitatively assess iron laydown morphology on the catalyst particle. The three images below show varying degrees of iron contamination on a catalyst particle. The first picture shows a fresh catalyst particle that is free of contaminant iron on its surface. The second picture shows a catalyst particle with a significant concentration of iron nodulation on the catalyst surface. The third picture shows a low boiling eutectic formed in the presence of an alkali metal like Ca or Na and is the severest form of iron poisoning.


David Hunt (Grace Davison)
Grace receives E-cat samples for most of the FCC units operating worldwide. The figure below shows the distribution of average equilibrium catalyst Fe levels for 2010 for all FCC units that have provided E-cat samples to Grace. Mean Fe levels are 0.57 wt% and the highest Fe level in one unit is 1.36 wt%.

Iron can be detrimental to the unit in many ways including bottoms conversion, catalyst circulation stability and SOx emissions.
Yaluris (1) showed using an Electron Probe Micro-Analysis (EMPA) technique that iron from organic iron sources is primarily a catalyst surface contaminant. Yaluris also used scanning electron microscopy and optical microscopy techniques to confirm Fe is a surface contaminant. The figure below is an EMPA image of an FCC catalyst particle cross section. Warmer colors on the surface of the particle confirm that Fe and CaO are primarily surface contaminants. EMPA Image of Two FCC Catalyst Particles

Yaluris (1) discussed how Fe contamination can lead to pore closure and nodule formation. The presence of Na and CaO can act as fluxing agents, aggravating the effect of Fe. The figure below shows Decant Oil or Main Fractionator bottoms yield vs. E-cat Fe plus CaO levels. Decant Oil increases at the higher contaminant levels due to the damaged catalyst pore structure.


Question 13: Severe fouling of diesel and gas oil hydrotreating preheat exchangers has been a growing problem. In your experience, what are the causes and how can these be prevented? Have you tried antifoulant injection in this service?
Dan Webb (Western Refining)
Fouling of the heat exchanger train is sometimes a problem particularly when processing cracked feed stocks. The fouling is often caused by polymer like compounds (gums) that form when petroleum distillates come in contact with air. When heated olefinic compounds react with absorbed oxygen to form gums that deposit in the preheat train.Iron scale and other particulates in the feed often adhere to these gums to produce severe fouling that restricts unit capacity and accelerates heat exchanger corrosion rates. Typically, every effort is made to avoid air ingress into any of the unit feed stocks. Fouling precursors may also be present in straight run feed stocks in the form of certain chemical contaminants that may be present in the crude or inadvertently introduced in an upstream process unit. Some precursors such as amines, carboxylic acids, and carbonyls form gums without air ingress into the feed. Antifoulants have been used successfully to mitigate fouling caused by these compounds in addition to mitigated fouling caused by oxygen contaminate cracked feed stocks.
Michael Chuba (Sunoco)
Typically distillate hydrotreaters exchanger fouling has been associated with cracked stocks that contain olefinic material and trace amounts of O2 coming in with the feed from tankage. In addition to oxygen-initiated polymerization, other impurities can lead to free radical formations that can promote polymerization reactions. These impurities include certain nitrogen and sulfur compounds well as some metal ions including iron, calcium, and magnesium.
In addition to free radial polymerization, condensation polymerization reactions can also result in fouling. In this route, two radicals can react to form a larger molecule. The new compound can continue to react and grow until it precipitates out of solution forming deposits.
What I would like to present here is an example of fouling we had on one of our units and how we have significantly reduce fouling via a simple jump over line.
Prior to conversion of this unit to ULSD the unit processed a mix of virgin and cracked distillate stocks. Historically this unit had exchanger fouling that was attributed to the presence of the cracked stocks. When the unit was converted to ULSD the cracked stocks were removed. The resulting feed was a 50:50 mix of direct rundown material from the crude unit and tankage. As a result of this change in operation it was anticipated that the fouling rate would decrease, however, during actual operation the fouling rate actually increased.
An initial program to address the problem included detailed analysis of the various feed stream followed by a targeted antifoulant chemical injection program. Results were somewhat effective but still left significant room for improvement. Continued investigation into the problem targeted O2 contamination coming from the material coming from tankage. The intermediate distillate tanks are cone roof design which would be relatively costly to convert to blanketed tanks. As a first step it was decided to install a jump over from the tank inlet line directly to the suction of the tanks’ transfer pumps. With this simple connection the average volume of material actually drawn from the tanks dropped dramatically.

This plot shows the impact on the heat transfer coefficient of the feed effluent exchanger as a result of this simple jump-over. The pink plot represents the previous cycle. At about ¼ of the cycle the jumpover line was installed. At this point significant fouling had already occurred. The discontinuity in heat transfer coefficient a week or two later was the result of a power failure. It is suspected that the rapid depressurization dislodges some of the fouling material thereby improving the heat transfer when the unit is re-streamed. This same response has been seen in previous emergency shutdowns. The green plot represents the current cycle which started with a clean set of exchangers and operation of the jumpover in service from day 1 of the cycle. As can be seen this simple jumpover has significantly reduced the rate of fouling compared to previous cycles. Since the only change was the potential ingress of O2 from the tank, this project confirmed the impact O2 had fouling.
Gregg McAteer (Nalco Company)
Fouling can be a serious problem in hydro-desulfurization (HDS) units because of their importance in producing fuels that should meet environmental specifications. Fouling can limit a unit's ability to maintain a specific feed rate or meet an extended turnaround date. It can greatly influence product quality as well as energy consumption, and catalyst or equipment life. Stricter limits on sulfur and aromatic content of finished fuels make fouling control even more important today. To achieve today’s limits of 0.05 wt.% for diesel, refiners must increase severity of refining operations, which often worsen fouling. Fouling ultimately necessitates shutdown and extensive maintenance, a costly process, both in terms of maintenance expenditures and lost production. Causes of fouling in diesel and gas oil hydrotreaters are both organic and inorganic in nature. The organic foulants are primarily gums formed as a result of processing cracked material and accelerated if the material is exposed to oxygen at any time. Antioxidants and/or antipolymerants are used to reduce the formation of gums and dispersants are used to keep any gums already formed from growing in size.
In one case an antifoulant program utilizing both an antioxidant and a dispersant was applied to a gas oil hydrotreater that normally fouled enough to require a shut down after an average of 440 days. The antifoulant program started on a fouled system and showed

a slight recovery of pressure drop. After a shutdown they started again and achieved a 1300 day run (see graphic below).
“Run 1” is shown in red and light blue. The red trend shows the steep increase in pressure drop during normal operation (without antifoulant program). The light blue trend shows the antifoulant program started, saw a small decrease in pressure drop, and then the unit was brought down for a regeneration. “Run 2” is shown as the dark blue trend and shows a lower fouling rate and longer run length with the antifoulant program. Customer estimated the ROI to be between 400-500%.
Phil Thornthwaite (Nalco Company)
Foulants typically found on the feed side of the preheat exchangers include various gums or polymers, iron sulphide and salts.
The organic fouling due to gums & polymers results from the polymerization of unstable species in the unit feed. The problematic species include olefins (generated in cracking processes), organic acids, mercaptans, ketones, aldehydes, phenols, organo-nitrogen and organo-sulphur compounds. Therefore, in order to determine the risk of organic fouling for a particular feed stream, detailed analysis for the problematic species can be useful guide in evaluating fouling propensity and mitigation strategies.
A typical level for concern for each problematic specie is outlined below:

Another key factor to consider is the oxygen content of the feed stream as this can promote the polymerization of various unstable compounds, particularly olefins. Therefore, it is good practice to exclude oxygen from feed storage tanks by ensuring tank seals and vents are in good condition and through the use of a nitrogen blanket. However, this method is ineffective with streams already exposed to oxygen since the nitrogen blanket will have no effect on oxygen reaction products such as aldehydes, peroxides and hydroperoxides.
Inorganic fouling is mainly caused as a result of iron sulphide that can either be carried from upstream units or generated in-situ in the preheat exchanger network. However, the latter is not so common since refiners choose the metallurgy to mitigate against sulphidic corrosion in most cases.
In order to mitigate and control fouling in the preheat train, chemical dispersants and antipolymerants are used. The properly selected dispersant will act upon the organic polymers by keeping them finely dispersed within the feed stream thus minimizing the risk of deposition on the exchanger surfaces. Likewise, dispersants can also prevent deposition of FeS by keeping them dispersed in the feed stream.
Antipolymerants act by disrupting the propagation and chain extending stages of the free radical polymerization reactions and by increasing the rate of termination. This will limit the rate of polymer growth within the preheat system. They will also minimize carbonyl formation which will in turn disrupt condensation polymerization reactions.
The key to monitoring the program effectiveness is through accurate monitoring of the preheat exchanger network. If the fouling results in a limitation of heat transfer efficiency, then a temperature survey of the exchanger network is carried out and this data is entered into a rigorous thermodynamic process model, such as Nalco’s MONITOR® program. This model will then use the plant data to calculate actual and normalized exchanger duties and heat transfer coefficients plus it will calculate the normalized furnace inlet temperature (NFIT). A successful antifoulant program will limit the decay in the NFIT and will generate significant returns for the refiner by improved energy efficiencies and optimized unit operation.
Robert Wade (ART)
We have not had success reducing fouling effects by adding antifoulants. It is our experience that adding antifoulants at best treats the symptom of the problem, and at worst further contributes to localized and downstream fouling. We recommend that the source of the fouling contaminant be identified through analysis and addressed at the source. If this is not possible then we revisit the basic design of the heat exchanger in question and ensure that it is operating in a shear controlled flow regime so that fouling effects are minimized
Question 41: Have the panel members considered 15% ethanol (E15) gasoline blending?
KOONTZ (HollyFrontier)
My first slide shows a little background. The EPA administers the Renewable Fuel Standard program that has volume requirements for renewable fuels. They established these volume requirements under the Energy Independence and Security Act of 2007. The EPA tracks compliance with the Renewable Identification Number (RIN) system, and they assigned a RIN to each gallon of renewable fuel.

HollyFrontier satisfies much of its requirement for conventional biofuel, which is essentially corn ethanol, by blending E10 gasoline at many of its terminals. Most of HollyFrontier’s gasoline is sold via pipeline to terminals owned by others; therefore, we are not able to supply our full mandated volume. HollyFrontier does purchase RINs from others. The decision to purchase ethanol to blend or the RINs is based on the economics of the cost of the RINs.

Ethanol blending for the refiner does have a significant impact on two critical gasoline properties: namely, octane and RVP. The hydrocarbon blend stock used for 90% of the E10product, which HollyFrontier calls sub-grade, has an octane rating of about 84. After blending with the 10% ethanol, the resulting octane is the regular 87. So being a refinery that adjusts total octane with its reformer severity, this allows us to run a lower severity, which is especially beneficial to those refineries with semi-regen reformers that operate at relatively high pressures and relatively low liquid volume product yield.

RVP is the other critical property affected by blending. When ethanol is blended with naphtha at a low concentration, the RVP of the gasoline is increased. Pure ethanol does have a low RVP; but when it is blended with hydrocarbon, it behaves more like a light hydrocarbon and actually raises the RVP. For example, with E10 for naphtha having an RVP of 9, the resultantE10 product has an RVP of about 10. So, to encourage ethanol blending, in 1990, the U.S. Congress passed a waiver known as the “One-Pound Waiver” which allows E10 gasoline to be sold at one psi (pound per square inch) higher than that normally required.
For the refinery, E15 would allow lower octane severity reformer operation, which would be beneficial. However, the EPA regulation implementing the “One-Pound Waiver” specifically references gasoline containing between 9% and 10% ethanol. The EPA has refused to extend this One Pounder Waiver to E15. Therefore, marketing E15 requires a sub-grade blendstock that has an RVP approximately 1 psi lower than normal gasoline sub-grade blendstock used for E10.

In addition, since January 2011, E15 has been permitted for use in light-duty motor vehicles manufactured after 2001. It was not approved to be used in small gasoline engines or other vehicles built before that due to concerns of material incompatibilities and corrosion. Furthermore, I have seen several places where current automobile manufacturers will not honor their warranties if the person used E15, even if the vehicle was manufactured after 2001. Also, the EPA requires that in order to sell E15 gasoline, a Misfuelling Mitigation Plan must be in place to prevent consumers from using the product in an unapproved engine. Today, there are very few retailers who have chosen to go through the additional trouble in order to sell the E15.
In conclusion, due to the absence of the “One-Pound Waiver” and the legal risk of corrosion or voiding the warranties of customers’ cars, HollyFrontier has chosen not to produce or blend E15.

SUBHASH SINGHAL (Kuwait National Petroleum Company)
Does the 15% have to do with the oxygen in the ethanol and other oxygenate like MTBE, or it is just because of the RVP limitations and other issues that you explained? From safety point of view, is there oxygen contained in the old oxygenate like ethanol? Is that one of the criteria for limiting the blending from 15% or 10%? Does this have to do with the oxygen
attached even though it is oxygenate?
KOONTZ (HollyFrontier)
My understanding, from reading, is that the E15 decision is not really based on logic. I think it was more of a U.S. Congress action. I do not really understand why they have not extended the “One-Pound Waiver” to E15. I do not think it is based on science.
KOONTZ (HollyFrontier Corporation)
The Environmental Protection Agency (EPA) administers the Renewable Fuel Standard (RFS) program with volume requirements for several categories of renewable fuels. EPA establishes the volume requirements for each category based on EISA (Energy Independence and Security Act of 2007) legislated volumes and fuel availability. EPA tracks compliance through the Renewable Identification Number (RIN) system, which assigns a RIN to each gallon of renewable fuel.
HollyFrontier satisfies much of its requirement for Conventional Biofuel (essentially corn ethanol) usage within RFS by selling E10 (10% ethanol) at many of its terminals. Most of HFs’ gasoline is sold via pipeline to terminals owned by others; therefore, to fully satisfy its mandated volume, HF purchases RINs from others. The decision to purchase ethanol from others and blend to E10 or to purchase RINs from others is based on economics.
Ethanol blending has a significant impact on two critical gasoline properties controlled by the refiner: octane and RVP. The hydrocarbon blendstock used for 90% of the E10 product (termed sub-grade by HF) has an octane rating of ~84. After blending with 10% ethanol (octane ~114) the resultant E10 octane is “regular” 87. For a refinery that normally adjusts reformer severity to satisfy the total gasoline pool octane, producing sub-grade allows for lower reformer severity and higher liquid yield. This improved yield is more pronounced for a semi-regeneration reformer that operates at relatively high pressure.
RVP is the other critical gasoline property affected by ethanol blending. When ethanol is blended with naphtha at low concentration, the RVP of the gasoline is increased. Even though pure ethanol has a low RVP [about 2 psia (pounds per square inch absolute)] due to O-H bonding, it behaves more like a hydrocarbon with a molecular weight of 46 when mixed with naphtha at low concentration. If ethanol is blended to 10% with 84 octane naphtha having an RVP of 9, the resultant E10 gasoline has an RVP of ~10. In order to encourage ethanol blending, the U.S. Congress passed the One-Pound Waiver in 1990 allowing E10 gasoline RVP to be 1 psi higher than that normally required by the EPA (One-Pound Waiver).
E15 would allow a refiner to produce an even lower octane sub-grade to blend with the ethanol and the RVP effect would be similar. However, the EPA regulation implementing the One-Pound Waiver specifically references gasoline containing between 9% and 10% ethanol. The EPA has refused to extend the one-pound waiver to E15. Therefore, to market E15 requires a sub-grade blendstock having an RVP over 1 psi lower than that required for E10.
Since January 2011, E15 has been permitted for use in light-duty motor vehicles manufactured after 2001. However, it is not approved for use in small engines and older vehicles due to concerns with material incompatibilities and corrosion. Furthermore, several automobile manufacturers will not honor their warranties if E15 gasoline was used in the vehicle (even for those manufactured after 2001). The EPA requires that in order to sell E15 gasoline, a Misfueling Mitigation Plan must be in place to prevent consumers from using the product in an unapproved engine. There are very few retailers who have chosen to get approval to sell E15.
Due to the absence of the One-Pound Waiver for RVP, the significant legal risk in selling a controversial product, and the minimal market demand HF has decided that it would be unwise to enter the E15 market at this time.
Question 91: What are the characteristics of FCC catalyst to minimize particulate emissions at the stack?
John Aikman (Grace Catalysts Technologies)
While there are several operational and mechanical factors that can influence a unit’s particulate emissions, the question asks specifically about the FCC catalyst; as such, the following discussion will address characteristics of fresh catalyst only.
There are four basic characteristics of FCC catalyst that can have direct effects on particulate emissions. These same characteristics will also affect particulate losses to the fractionator and slurry product. The first characteristic is simply the amount of fines content coming into the unit with the fresh catalyst due to the manufacturing process. Figure 1 is an example of a typical fresh catalyst particle size distribution, with a theoretical depiction of a cyclone’s ability to retain fresh catalyst particles. DPTh is the smallest particle diameter which can reliably be collected by a cyclone and is used to model cyclone performance. Particles below this size will be lost by the cyclone.
A review of the Grace Ecat database showed that none of the FCCU’s in North America can retain any 0-20 μ range particles. In addition, they only retain an average of approximately 4.0 wt% in the 0-40 μrange. Fresh catalyst typically ranges anywhere from 9 to 16% of 0-40 μ depending on the supplier andmanufacturing process. Some units require higher amounts of 0-40 μ range particles to help with circulation.

The next characteristic of fresh catalyst that must be considered is the particle density. he DPTh mentioned above will decrease with increased catalyst particle density, per Equation 1 below. This means that cyclones can retain smaller particle sizes as the particle density increases. This is due to the centrifugal force acting on a heavier particle. However, particle density is not the same as apparent bulk density (ABD). Industry typically measures and reports ABD as part of the routine Ecat analysis, but this should not be mistaken for particle density for cyclone efficiency purposes. Since Al2O3 is denser than SiO2, catalysts with higher alumina content will have higher catalyst particle density.

The third characteristic is the inherent attrition resistance of the fresh catalyst. Industry measures the attrition resistance via a variety of tests, with the primary goal of providing a relative indication of catalyst attrition resistance. Grace utilizes the DI test or Davison Index. On the DI scale, a lower number is less likely to cause attrition and generate microfines. It is usually not valid to compare attrition resistance results obtained from different laboratories. Additionally, it is important to note that the energy applied to a catalyst sample during attrition testing is much more severe than commercial conditions.
As discussed above, the majority of the microfines created in the FCCU will leave the unit through either the reactor or regenerator cyclones, with the latter potentially contributing to increased particulate emissions at the stack.
'The attrition resistance of the catalyst is a function of the manufacturing process and the binder material utilized during the manufacturing process. Figure 2 is an example of how a refiner improved the FCCU stack opacity with catalyst formulation. The reduction was achieved changing to a Grace supplied catalyst with lower DI and lower 0-40 μ content in the fresh catalyst.

The final characteristic of fresh catalyst that affects particulate emissions is its morphology. Morphology can be defined as the study of the form and structure of a particle and its specific structural features. A catalyst particle that has a smoother exterior surface is less likely to generate microfines in an FCCU. Even catalysts with a low fresh DI measurement can cause increased particulate emissions if there are surface irregularities resulting from the manufacturing process. In order to demonstrate this visually, Figures 3 and 4 present SEM’s (scanning electron microscopy) of “bad” and “good” fresh catalyst morphology for a side-by-side comparison.
Figure 3 and 4 SEM’s of Fresh Catalyst (magnified X250)
“Bad Morphology” “Good Morphology” In conclusion, there are several characteristics of fresh catalyst that can be controlled to reduce particle losses and thereby reduce flue gas emissions. Specifically, to lower emissions the fresh FCCcatalyst should possess the following characteristics: a particle size distribution with an optimal range of 0-40 μ particles, higher catalyst particle density, lower DI, and superior morphology. Grace’s alumina-sol technology provides superior binding to the catalyst particle leading to best-in-industry attrition resistance. The versatility and performance of alumina-sol catalysts coupled with Grace’s manufacturing capability, have resulted in wide-market acceptance and as a result, Grace is the preferred FCC technology for loss sensitive units around the world.
Question 65: What are the impacts on coker operation (yields, capacity, energy, coke quality) of FCC slurry oil in the feed?
Gary Gianzon (Marathon Petroleum Company)
When one of MPC’s refineries starts processing heavy Canadian resid, they add 5 to 10 volume percent of slurry oil in the feed to mitigate making shot coke. The slurry also helps meet anode grade specifications on metals and sulfur. Processing slurry backs out resid processing which can impact unit economics.
FCCU slurry has a similar boiling range to heavy coker gasoil, so a large amount of slurry flashes out of the drum and ends up in the heavy coker gasoil product. The coke yield from slurry feed is around 2 to 3 x Concarbon (depending on coker unit operation) which is significantly higher than vacuum resid at 1.3 to 1.6 x Concarbon. If a high percentage (over 10 percent) of slurry is processed in the coker unit, the slurry can cycle up between the coker and FCCU unit. The amount of recycle built-up is somewhat self-correcting depending on operations in the coker and FCC and whether the HCGO is processed in a FCCU Feed Hydroteater.
Rajkumar Ghosh (Indian Oil Corporation)
We are adding approx. 3–4 wt% FCC Slurry oil in Coker feed in one of our Coker and about 10 wt% in another. We also had undertaken a study in the Delayed Coker pilot plant in our R&D centre. Our experiences with processing of FCC slurry oil in the Coker feed, based on field and pilot plant results, are as under:
a) Yield: The impact of slurry oil in Coker feed depends upon the quality of the base feedstock, CLO/slurry oil and also the pressure / temperature of the coke drums. If FCC slurry oil boiling point distribution and the coke drum pressure / temperature are such that most of the slurry oil vaporizes out of the coke drum, yield of coke and gas reduces with increase in distillate yield.
In case of Fuel grade Coker, with CLO (with minimum overlap of LCO) below 10wt% in VR feed, coke yield by and large may be constant or may increase marginally depending on the relative quality of VR and CLO. Yields of total gas and liquid decrease marginally. Beyond 10 wt% (10-20 wt%) of CLO in VR feed, the coke yield may increase up to 4 wt%.
b) Capacity: The Coke produced with significant FCC slurry in Coker feed (>10 wt%) has a close-knit Coke matrix which ensures good porous structure to the Coke bed. This reduces the chances of hot spots and blowouts. But the negative impact of adding FCC slurry is pronounced where the coke drum is already limiting, as the porous structure results in lower coke bed bulk density and hence lesser vapor space in the Coke Drum. It may limit the Coker capacity.
c) Product quality: Tendency of formation of Shot coke significantly reduces with the addition of FCC slurry in the Coker feed, as it keeps asphaltenes in solution form. As per our experience at Panipat Coker, impact of slurry addition in the Coker feed is clearly visible on the Coke quality w.r.t. reduction in Shot coke formation. With increased FCC slurry in Coker feed, increase in Silica content in the green Coke would be a criterion to limit its wt% in the feed. This is significant for the Cokers producing Anode grade coke. Typical limit of Silica in Anode grade green Coke is 0.02 wt % max. Depending on the quality of the slurry oil and unit operating conditions, there may be a negative impact on the quality of the LCGO and HCGO. They will become more aromatics and heavier.
d) Energy: Slurry processing will require higher heater duty. High aromatic content in the slurry oil prevents the precipitation of Asphaltenes and thus increases the heater run length. Injection of slurry oil into the coke feed is limited by refinery configuration. In our Refineries with FCC and/or Hydrocracking units, we limit the slurry oil within 5 to 10 wt% on fresh feed to Coker. Increase in injection rate can lead to a massive recycle between the Coker and the FCC or will result in accelerated catalyst deactivation in the Hydrocracker unit.
Eberhard Lucke (Commonwealth E&C)
In general, FCC Slurry has a similar effect as VGO in terms that it replaces residue in the feed and increases mainly the HCGO yield. The difference in this case is that FCC Slurry is a highly aromatic stream and is often used as additional Coker feed (up to 15wt% max. recommended) to reduce heater fouling and to push coke morphology to sponge coke (for anode grade coke). The heavy aromatics in the FCC Slurry help keeping asphaltenes in solution a lot longer and promote coke formation by poly-condensation, therefore increasing sponge type coke content in the coke bed (preferred for low sulfur, anode grade coke production). On the downside, FCC Slurry will contain entrained catalyst fines and – if too high in concentration – may have a negative impact on fouling rates in the charge heater(s). The fine catalyst particles can deposit inside the heater tubes, act as seeds for coking and may promote deposits of heavy oil and coke fines from the oil film inside the tubes.




